Question 1 The following figure shows a(an): A) d orbital B) p orbital C) s orbital D) forbital Question 2 Which of the following elements correctly belong to the s-block of the electron configuration Table? Atomic number 11 12 13 14 15 16 17 18 A) Phosphorus, Sulfur B) Sodium, Magnesium C) Chlorine, Argon D) Aluminum, Silicon Element Na ✓ Mg Al Si P S Cl Ar Orbitals 1s 2s ↑↓ ↑↓ T↓ T↓ 2p ↑↓↑↓↑↓↑↓ T ↑↓↑↓↑↓↑ ↓ ↑↓ ↑↓↑↓↑↑↓ TT 3s TL TLT↓↑↓↑↓↑ ↑↓↑↓↑↓↑↓↑↓↑ TIAL 3p ↑ ↑↑ ↑ ↑↓ T↓ ↑↓↑↓↑↓↑↓↑↓↑ ↑↓ T↓ T↓↑↓↑↓ T↓ ↑↓↑↓↑ T↓ T↓ ↑↓↑↓↑↓ TL T↓ T ↓ T ↓ Electron configuration 1s²2s²2p 3s¹ 15²23²2p3s² 15²2²2p 3s 3p¹ 1s 2s 2p 3s 3p² 1522s²2p 3s 3p³ 15²2s²2p 3s 3p 1s 2s 2p 3s 3p 1s 2s 2p 3s 3p
Question 1 The following figure shows a(an): A) d orbital B) p orbital C) s orbital D) forbital Question 2 Which of the following elements correctly belong to the s-block of the electron configuration Table? Atomic number 11 12 13 14 15 16 17 18 A) Phosphorus, Sulfur B) Sodium, Magnesium C) Chlorine, Argon D) Aluminum, Silicon Element Na ✓ Mg Al Si P S Cl Ar Orbitals 1s 2s ↑↓ ↑↓ T↓ T↓ 2p ↑↓↑↓↑↓↑↓ T ↑↓↑↓↑↓↑ ↓ ↑↓ ↑↓↑↓↑↑↓ TT 3s TL TLT↓↑↓↑↓↑ ↑↓↑↓↑↓↑↓↑↓↑ TIAL 3p ↑ ↑↑ ↑ ↑↓ T↓ ↑↓↑↓↑↓↑↓↑↓↑ ↑↓ T↓ T↓↑↓↑↓ T↓ ↑↓↑↓↑ T↓ T↓ ↑↓↑↓↑↓ TL T↓ T ↓ T ↓ Electron configuration 1s²2s²2p 3s¹ 15²23²2p3s² 15²2²2p 3s 3p¹ 1s 2s 2p 3s 3p² 1522s²2p 3s 3p³ 15²2s²2p 3s 3p 1s 2s 2p 3s 3p 1s 2s 2p 3s 3p
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter7: The Structure Of Atoms And Periodic Trends
Section: Chapter Questions
Problem 64GQ: Answer the questions below concerning ground state electron configurations. (a) What element has the...
Related questions
Question
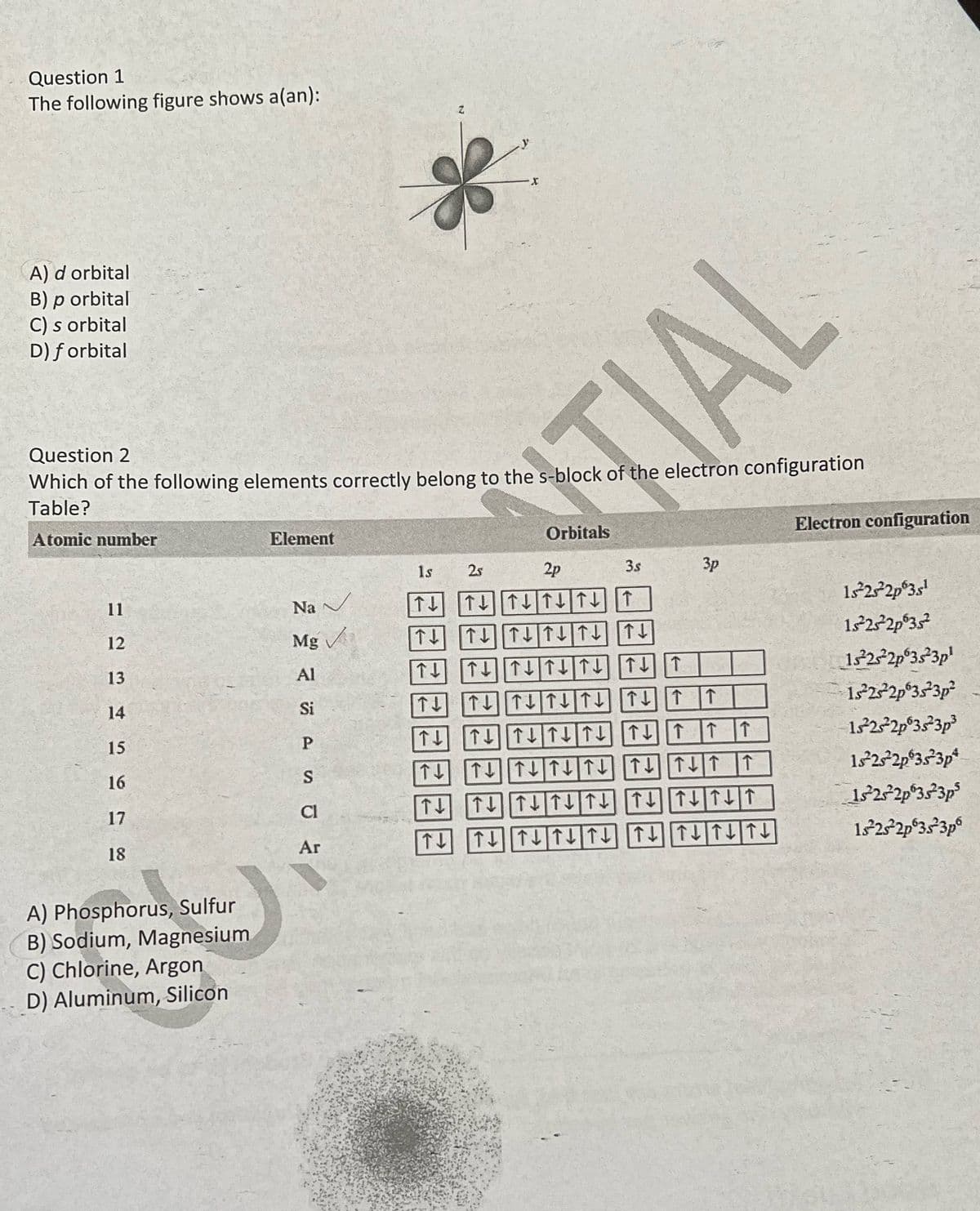
Transcribed Image Text:Question 1
The following figure shows a(an):
A) d orbital
B) p orbital
C) s orbital
D) forbital
11
12
13
14
15
16
17
18
Question 2
Which of the following elements correctly belong to the s-block of the electron configuration
Table?
Atomic number
A) Phosphorus, Sulfur
B) Sodium, Magnesium
C) Chlorine, Argon
D) Aluminum, Silicon
Element
Na
Mg✓✓
P
Z
Ar
X
TIAL
25
Orbitals
1s
2p
↑↓ ↑↓↑↑↓↑↓↑
↑↓↑↑↓↑↓↑↓↑↓
3.s
3p
↑↓ ↓↑↓↑↓↑↑↓↑
T↓
T↓ ↑↓↑↓↑↓↑↓↑
T↑↓
↑↓↑↓↑↓↑↓↑↓↑↑↑
↑↓
↑↓↑↓↑↓↑↓
↓↑↓↑↑
↑↓ T↓ T↓↑↓↑↓ T↓ ↑↓↑↓↑
↑↓↑↓↑↓↑↓ ↑↓↑↓↑↓ ↑ ↓
↑
Electron configuration
1s²2s²2p 3s¹
15²23²2p635²
1322²2p038²3p¹
15²25²2p 3s 3p²
18²2²2p 3s 3p³
18²2s²2p 3s 3p*
1s 2s 2p 3s 3p
18²2s²2p 3s 3p
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
