QUESTION 12 Heptane (C7H16) is a hydrocarbon fuel used for gas barbecues. Balance the chemical equation (given below) for the reaction that occurs when heptane burns in air. What are the coefficients in the balanced equation? C7H16 + 02 CO2 + H20 O 1, 11, 7, 8 O 1, 15, 7, 16 O 1, 22, 7, 8 O 1, 1, 7, 8 QUESTION 13 Acetylene (C2H2) burns in pure oxygen with a very hot flame which is used in welding and cutting metals. The chemical equation for the reaction is the following: 2 C2H2 + 5 02→ 4 CO2 + 2 H20. How many moles of oxygen are required to burn 3.4 mol of acetylene? Type only the number. Click Save and Submit to save and submit. Click Save All Answers to save all answers. NOV 53 6.
QUESTION 12 Heptane (C7H16) is a hydrocarbon fuel used for gas barbecues. Balance the chemical equation (given below) for the reaction that occurs when heptane burns in air. What are the coefficients in the balanced equation? C7H16 + 02 CO2 + H20 O 1, 11, 7, 8 O 1, 15, 7, 16 O 1, 22, 7, 8 O 1, 1, 7, 8 QUESTION 13 Acetylene (C2H2) burns in pure oxygen with a very hot flame which is used in welding and cutting metals. The chemical equation for the reaction is the following: 2 C2H2 + 5 02→ 4 CO2 + 2 H20. How many moles of oxygen are required to burn 3.4 mol of acetylene? Type only the number. Click Save and Submit to save and submit. Click Save All Answers to save all answers. NOV 53 6.
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter3: Chemical Reactions
Section3.2: Balancing Chemical Equations
Problem 1RC: The (unbalanced) equation describing the oxidation of propanol is 2 C3H7OH(l) + __O2(g) 6 CO2(g) +...
Related questions
Question
answer questions 12 and 13 and 14 and 15
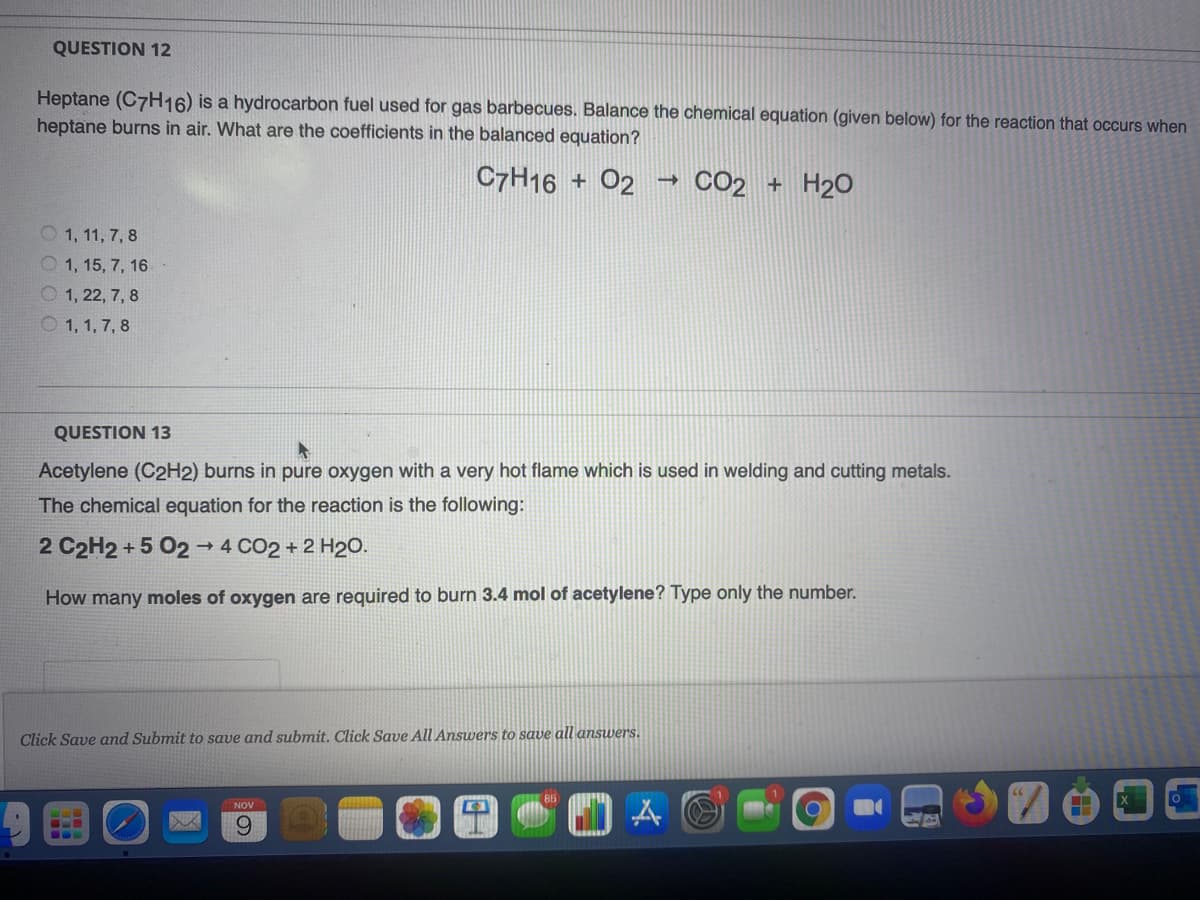
Transcribed Image Text:QUESTION 12
Heptane (C7H16) is a hydrocarbon fuel used for gas barbecues. Balance the chemical equation (given below) for the reaction that occurs when
heptane burns in air. What are the coefficients in the balanced equation?
C7H16 + 02
CO2 + H20
O 1, 11, 7, 8
O 1, 15, 7, 16
O 1, 22, 7, 8
O 1, 1, 7, 8
QUESTION 13
Acetylene (C2H2) burns in pure oxygen with a very hot flame which is used in welding and cutting metals.
The chemical equation for the reaction is the following:
2 C2H2 + 5 02→ 4 CO2 + 2 H20.
How many moles of oxygen are required to burn 3.4 mol of acetylene? Type only the number.
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
NOV
6.
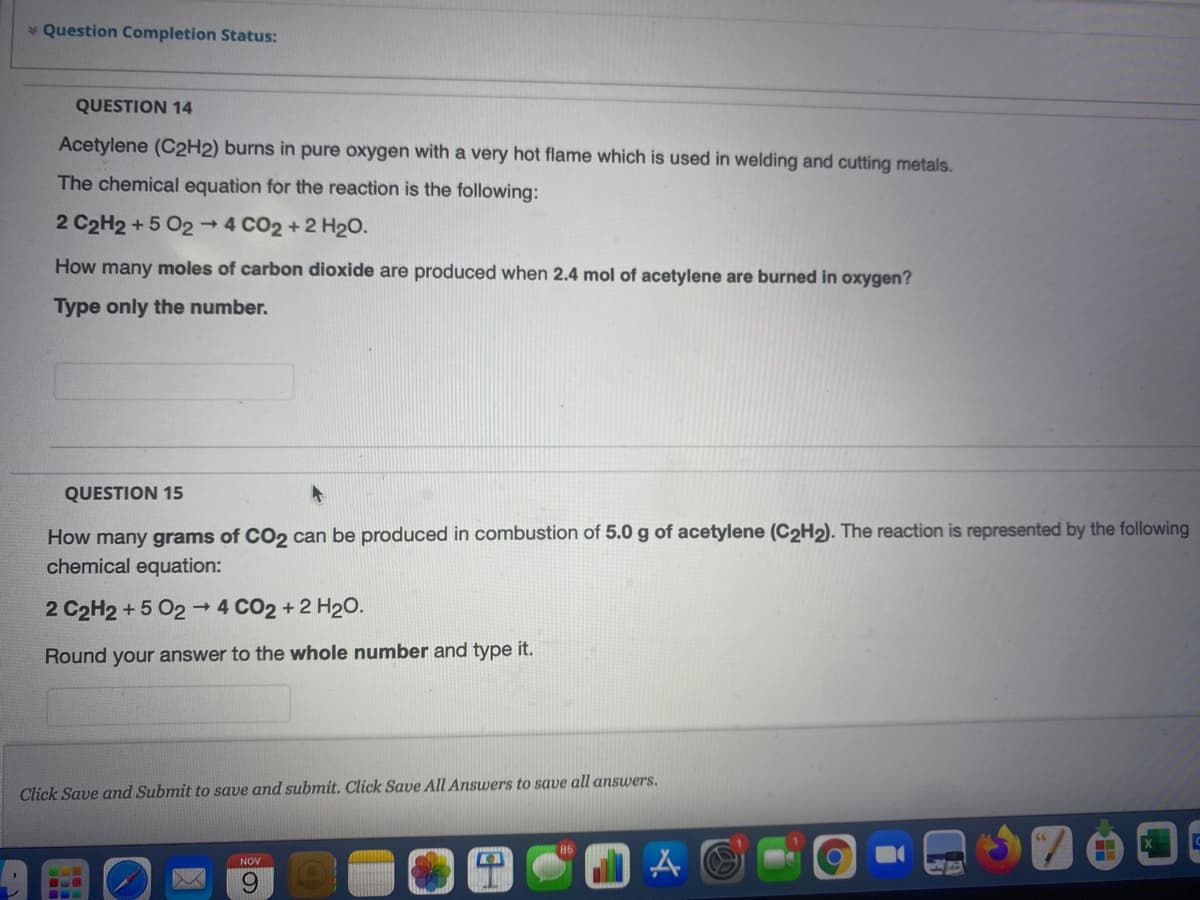
Transcribed Image Text:* Question Completion Status:
QUESTION 14
Acetylene (C2H2) burns in pure oxygen with a very hot flame which is used in welding and cutting metals.
The chemical equation for the reaction is the following:
2 C2H2 +5 O2 4 CO2 +2 H2O.
How many moles of carbon dioxide are produced when 2.4 mol of acetylene are burned in oxygen?
Type only the number.
QUESTION 15
How many grams of CO2 can be produced in combustion of 5.0 g of acetylene (C2H2). The reaction is represented by the following
chemical equation:
2 C2H2 + 5 02→
4 CO2 +2 H20.
Round your answer to the whole number and type it.
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
NOV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning