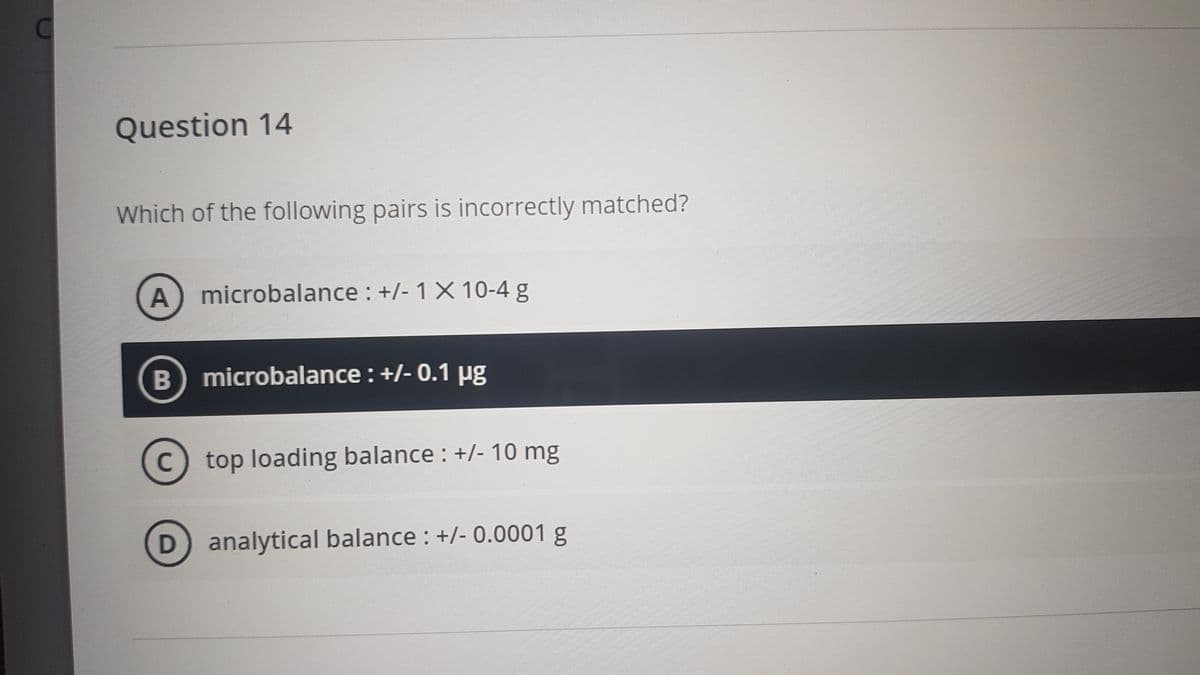
Question 14 Which of the following pairs is incorrectly matched? A microbalance : +/- 1X 10-4 g B microbalance: +/- 0.1 ug top loading balance : +/- 10 mg analytical balance : +/- 0.0001 g
Q: 2 CO(g) + O2(g) +2 CO2(g) K= 6.1x 105 If [CO] = 0.00100 M and [O2] = 0.00127 M, determine [CO2], in ...
A:
Q: Sulfur dioxide combines with O2 in the presence of a catalyst as represented by the equation 2SO2(8)...
A:
Q: 1. Which action between Mg ribbon and HCI will NOT increase the rate of reaction? A. Increase the te...
A: Here we have to determine the factor that increase rate of reaction between Mg ribbon and HCl.
Q: 3-38 Give IUPAC names for the following compounds: (a) CH3 (b) CH3 (c) H3C CH3 CH3CHCH2CH2CH3 CH3CH2...
A: According to IUPAC nomenclature first we have to select the longest chain of hydrocarbon which conta...
Q: Calculate the pH for each of the cases in the titration of 25.0 mL of 0.180 M pyridine, C,H, N(aq) w...
A: Given, Molarity of C5H5N = 0.180 M Volume of C5H5N = 25.0 mL Molarity of HBr = 0.180 M Base diss...
Q: Nal(sq) + Hg,(C,H,O;)a(aq) → Express your answer as a chemical equation. Enter NOREACTION I no react...
A:
Q: 4NH3 (g) + 502 (2) 4NO(R) 6H20 (g) --> Answer each question that follows. O a. Increasing the pressu...
A: Given : We have to pick the correct statement for the given reaction.
Q: A plot of Gibbs Energy against the extent of reaction has been constructed. Which point in the graph...
A:
Q: What is the product of the nucleophilic substitution reaction shown below? CH;SH HBr HS H SH Multipl...
A:
Q: Consider the reaction A+B+CD The order of the reaction with respect to each reactant is given below:...
A: Given the order of the reaction with respect to A, B and C are respectively 1, 1, and 0. Hence the ...
Q: 1. Catalyst in a living organism is referred as: A. Catalytic Converter В. Масгomolecule C. Biomolec...
A: 1) Catalysts of living organisms.
Q: (SPM10-29] Which equations represent double decomposition reactions that form a precipitate? Na So. ...
A: Given reactions are : I. CuSO4 + Na2CO3 -------> CuCO3 + Na2SO4 II. CuSO4 + Mg(NO3)2 ------->...
Q: At a certain temperature, the equilibrium constant K for the following reaction is 3.11 × 10¯°: H,(g...
A: We have to predict the equilibrium composition , equilibrium constant value.
Q: An unknown hydrocarbon is subjected to elemental analysis. The results of this test show that the co...
A:
Q: 6. If 0.025 mol of COCI2 (g) is placed in a 1.0L container at 400°C, 16.0% of the COC12 (g) is disso...
A: KC is equilibrium constant
Q: Which of the following factors affects the equilibrium of reversible reactions involving gas? ...
A: To identify: The factors that affect the equilibrium of reversible reactions involving gas.
Q: at a certain temperature this reaction follows second-order kinetics with a rate constant of 47.7 M ...
A: Reaction:- 2HI ------> H2 + I2
Q: Was there any physical change in the characteristics of trichloroethane when it was added to water?
A: Trichloroethane is micible in water. When it is added to water there is no any physical change.
Q: A rock is placed on a balance and its mass is determined to be 20.140 g. When thesame rock is placed...
A: • The values provided in the question are:- i) Mass of a rock, m = 20.140 g ii) Initial volume of ...
Q: There are two possible tautomers for histamine. Which one is more stable and why? HN- NH3 NH3 A
A: The two tautomers given are,
Q: For the reaction Br2(g) + Cl2(g) 2BRCI(g), at equilibrium, it is found that the concentrations of Br...
A: Here given [Br2]=0.398 M, [Cl2]=0.351M, [BrCl]=2.05×10-5M FFrom the given equation Kc=[BrCl]2/[Br2][...
Q: Question 9 What is the cell potential for the following reaction at 25 *C? Ni(s) + Cu2*(0.010 M) – N...
A: This question is related to electrochemistry. Electrochemistry is branch of chemistry in which we de...
Q: In the procedure you followed for the above experiment, the pipet that was used to measure the unkno...
A:
Q: Be sure to answer all parts. Cinnamic alcohol is used mainly in perfumery, particularly in soaps and...
A:
Q: 7)ln molecules below, circle the group that showsthe most down fieldin both 'H and C NMR shift. CH3-...
A: 7. A molecule is given below CH3-CH2-CH2-O-CH(CH3)2-CH2(Cl)-CH=CH-CH3. We have to indicate the most ...
Q: Br 1. PPH3 2. n-butyllithium H. 3.
A: Wittig reaction is the reaction which is used to produce alkene by the help of carbonyl compound. Tr...
Q: What is the appropriate size of volumetric flask to use if you want to dilute a 75.00 mL solution of...
A:
Q: A first order reaction is found to have a half- life of 1.2 seconds. This reaction is initiated with...
A: The rate constant (k) for a first-order reaction can be calculated from the half-life time t12. It c...
Q: QUESTION 4 Which of the following compounds cannot hydrogen bond? O CH3NH2 OCH3CH(OH)CH3 O (CH3CH2CH...
A:
Q: Name Haley oyle 20yle Sec. QUESTIONS 1. Why must the Erlenmyer flask and graduated cylinder be dried...
A: The answer are as follows:
Q: NO2(3) + CO(g) –→ NO(g) + CO2(g) The rate constant at 699 K is measured to be 2.62 Ms1, and at 896 K...
A:
Q: The cell notation for the Galvanic cell is Cd(s)Ca2*/12(s)/" The Standard Reduction Potential are as...
A:
Q: A sample of 1.00 mol of perfect gas molecules with Cp = 7/2 R is put through the following cycle: (a...
A:
Q: What are the AHrxn and ASrxn values that indicate a spontaneous reaction at all temperatures? O AHrx...
A:
Q: What is the packing efficiency (in %) for a simple cubic unit cell? Give answer with three decimal p...
A:
Q: At 400 K, an equilibrium mixture of H2, 12, and HI consists of 0.065 mol H2, 0.079 mol 12, and 0.13 ...
A:
Q: In a first-order reaction, the half-life is 137 minutes. What is the rate constant? O 1.22 x 10-4 s-...
A: The rate constant and half life of first order reaction is related as follows - Half-life = 0.693/k
Q: General Problems 3-48 For each of the following compounds, draw an isomer that has the same function...
A: The compounds given are,
Q: Acetal product formation requires an acid catalyst in the second step, but NOT in the first step. T...
A: We have to predict whether given Statement is true or not.
Q: A 50.0 mL solution of 0.139 M KOH is titrated with 0.278 M HC1. Calculate the pH of the solution aft...
A:
Q: 15.0 L of an ideal gas at 298 K and 3.36 bar are heated to 350 K with a new pressure of 3.80 bar. Wh...
A:
Q: The molar conductivity at infinite dilution of hydrobromic acid, sodium acetate, & sodium bromide at...
A: Molar conductivity at infinite dilution of : Hydrobromic acid (HBr) = 520.1 S cm2 mol-1 Sodium acet...
Q: a) CH3-CH2 CH2-CH3 b) CH3 -CH2-CH=CH-CH2-CH3
A: Step1- first identify the longest carbon chain. Step2- start numbering from that side such that sen...
Q: 1. The positivity rate in a certain group is 3.5%. Determine the probability that a batch will test ...
A:
Q: please solve for part a & b .. im really lost and don’t know how to go about it , thank you!
A: We have to find out the ∆H,∆S and Tb, Tf for the following reaction .,
Q: Question 26 Why are some metabolic reactions coupled to the hydrolysis of ATP? To bypass metabolic r...
A: The correct option is:
Q: What is the shape of OXEF4
A: A molecule is given is XeOF4. We have to tell its shape. We know that hybridization is H = Number o...
Q: The standard cell potential for the reaction at 298.15 K: Fe (s) + Zn2+ (aq) ⇋ Fe2+ (aq) + Zn (s) ...
A:
Q: What is the cell potential for the following reaction at 25 °C? Ni(s) + Cu2+(0.010 M) → Ni2+(0.001...
A:
Q: Which of the following is/are TRUE about Galvanic cell? I. The Gibbs Free Energy of the cell is nega...
A: A Galvanic cell is an electrochemical cell, in which the chemical energy is converted into electrica...
Step by step
Solved in 2 steps with 1 images

- I got a task in analytical chemistry to calibrate, standardize, verify and validate an analytical balances, can you help me?I got a task in analytical chemistry to calibrate, standardize, verify and validate an analytical balancesidea how gravimetric analysis provides accurate results despite of it being time consuming
- Using a 10mL pipette, you prepare a standard solution by pipetting 10.00mL of the standard iron solution [0.25g/L] into a 100mL volumetric flask. Then, using a 25mL pipette you transfer a 25mL aliquot of this solution into a 250mL volumetric flask and dilute up to the mark. Calculate the % uncertaintyUsing a 25mL pipette, you prepare a standard solution by pipetting 25.00mL of the standard iron solution[0.25g/L] into a 500mL volumetric flask. Then, using a 10mL pipette you transfer a 4mL aliquot of this solution into a 50mL volumetric flask and dilute up to the mark. Calculate the % uncertainty.Q4 (a) Analyze the importance of dropping mercury electrode on polarography. (b) (i) Calculate the molarity of MgSO4 in a solution containing 1.5 g ofMgSO4 in a volume of 250 mL?(ii) Calculate the weight of MgSO4 in grams which is present in 100mL of 0.076 M MgSO4?(c) Evaluate the key differences between paper chromatography and gaschromatography
- A patient has a deep vein thrombosis (DVT) and needs to be given tinzaparin at a dose of 175units/kg. The patient weights 68 kg. What dose does he need? The strengh(concentration) of tinzaparin available is 20000 units/mL. How many mL should the patient receive?(show work)Question 2 (b) Name and discuss five stages in which gravimetric analysis procedure may be dividedA 50.00 (±0.03) mL portion of an HCl solution required 29.71(±0.03) mL of 0.01963(±0.0030) M Ba(OH)2 to reach an end point with bromocresol green indicator. The molar concentration of the HCl is calculated using the equation below (attached image): a.) Calculate the uncertainty of the result (absolute error). M=0.02333(±?????) M b.) Calculate the coefficient of variation for the result. CV= (Sy/y) x 100%
- Give typed full explanation After heating 1.1929g CaCO3 and MgCO3 for some minutes the mass of the mixture decreased to 0.8015. The %comp of CaCO3 turned out to be 54.29% and MgCO3 %comp is 45.71%. Calculate the percent error/percent yield.Cat Noir was tasked to determine the concentration of a CuSO4 unknown solution. She prepared 5 different standards of CuSO4 using the table below as her guide. She ran the standards and the unknown solution in the UV-VIS Spectrophometer, and recorded the absorbances of each solution. Show linear equation derived from calibration curve and solution to concentration of the unknown solution below. Encircle final answer. Cuvette Volume 0.5000 M CuSO4 Volume distilled H2O Absorbance 1 1.000 mL 4.000 mL 0.607 2 2.000 mL 3.000 mL 0.655 3 3.000 mL 2.000 mL 0.709 4 4.000 mL 1.000 mL 0.797 5 5.000 mL 0.000 mL 0.832 Blank 0.000 mL 5.000 mL 0.000 Unknown solution 0.684Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer) MW: Cu (63.55) a) 2.5 ppb Cu2+ b) 2,500,000 ppb Cu c) 2.5 ppt Cu2+ d) 39.34 mM e) 0.03934 M f) 0.07868 N (in precipitation reaction) g) 0.07868 N (in redox into Cu+)



