Question 17 5 pts Molten sodium chloride can be electrolyzed to form liquid sodium metal and chlorine gas. The half-reaction at the anode is shown: 2 CI (1) – Cl2(g) + 2 e What volume of chlorine gas, in L, is generated by the application of a 15 A current for 1.25 hrs at a temperature of 298K and a pressure of 1 atm? Report your answer to two decimal places and do not include units with your answer.
Question 17 5 pts Molten sodium chloride can be electrolyzed to form liquid sodium metal and chlorine gas. The half-reaction at the anode is shown: 2 CI (1) – Cl2(g) + 2 e What volume of chlorine gas, in L, is generated by the application of a 15 A current for 1.25 hrs at a temperature of 298K and a pressure of 1 atm? Report your answer to two decimal places and do not include units with your answer.
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 60E
Related questions
Question
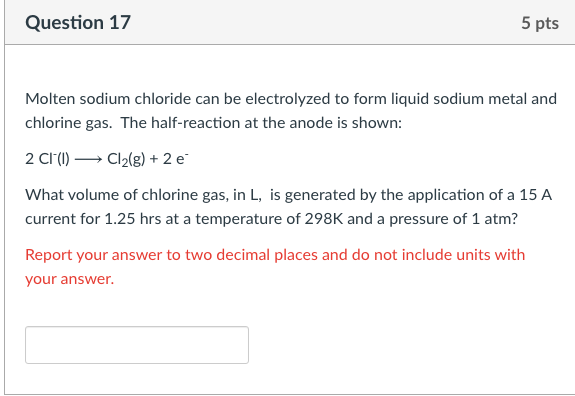
Transcribed Image Text:Question 17
5 pts
Molten sodium chloride can be electrolyzed to form liquid sodium metal and
chlorine gas. The half-reaction at the anode is shown:
2 Cl(1) → Cl2(g) + 2 e
What volume of chlorine gas, in L, is generated by the application of a 15 A
current for 1.25 hrs at a temperature of 298K and a pressure of 1 atm?
Report your answer to two decimal places and do not include units with
your answer.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
