Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
Transcribed Image Text:101 Chem101
b My Questions | bartleby
x +
->
app.101edu.co
M
Apps
G
M Gmail
YouTube
Maps
a AMAZON
Translate
O Gflights
Case Status Onlin...
b Homework Help a...
C Get Homework He... > KATAPULK CUBA
23 Agencia Supermar..
Reading List
Question 3 of 22
Submit
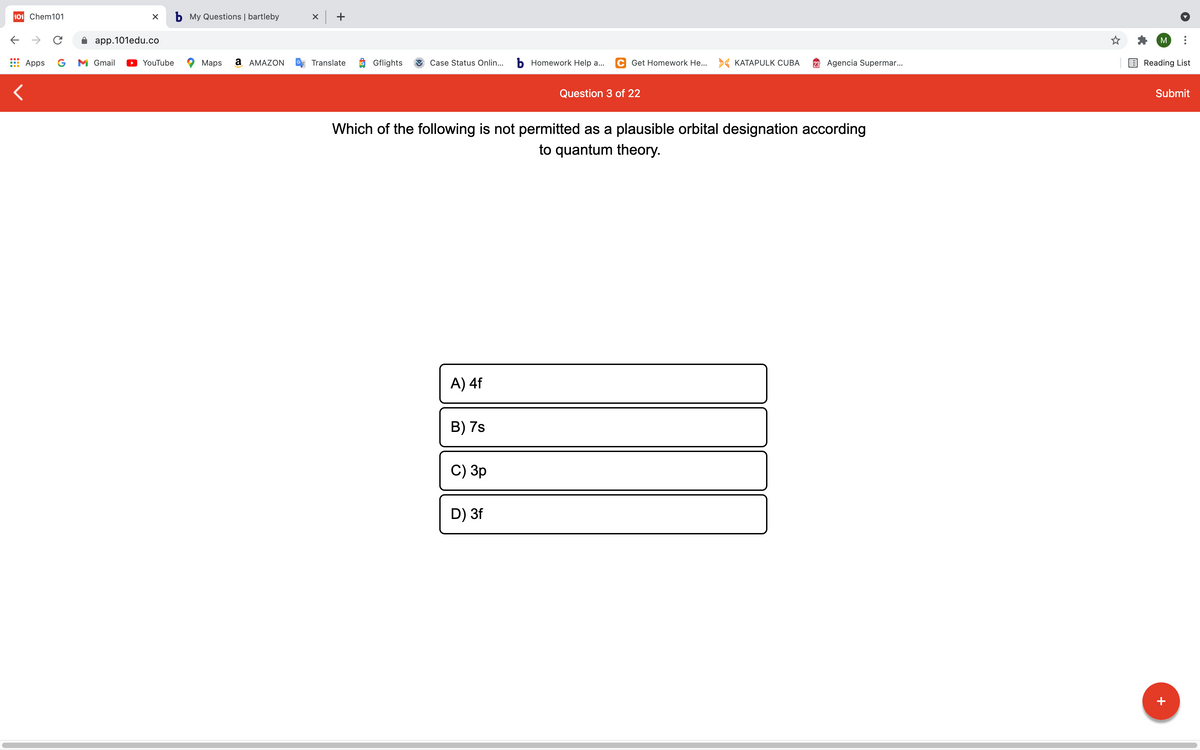
Which of the following is not permitted as a plausible orbital designation according
to quantum theory.
A) 4f
B) 7s
C) Зр
D) 3f
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you



