Q: Question #3 A 26.8 L tank of nitrogen gas is at 25.0 °C and 2.2 atm. If the temperature stays at…
A:
Q: Predict the product of the following reaction conditions and draw a mechanism to illustrate its…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Predict the major products of this organic reaction.
A: Major product for the given reaction is 2-methylcyclohexanoneExplanation:Step 1:- oxidation of…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. NaOEt, room temperature 2.…
A: The objective of the question is to draw the major product of the given reaction.
Q: Which is correct about this reaction under standard condition? O || O II O-P-O-P-O-P-OCH₂ "O "O O O…
A: Given is organic reaction.The given starting compound is Adenosine triphosphate.(ATP)The given…
Q: out what the highest order Cn and Sn axis of rotation are). Be very clear and neat. a) (1 point).…
A: The objective of the question is to identify the highest order of rotation axis (Cn) and improper…
Q: What changes in the IR spectra would be expected if the conversion of the carboxylic acid to the…
A: Given:Carboxylic acid converted into esterWe have to find the changes in the IR spectra that can be…
Q: (5) Draw the most stable chair conformation of the cyclohexane shown below. CH3 CI H3C Br OH "CH 3
A: Chair confirmation is the most stable conformer of cyclohexane.The chair with the least number of…
Q: What is the order of the reaction A → B + C when a graph of ln[A] versus time gives a straight line…
A: The objective of the question is to determine the order of the reaction A → B + C based on the…
Q: Give detailed Solution with explanation needed..don't give Handwritten answer..don't use Ai for…
A: The objective of the question is to identify the species that is oxidized and the species that is…
Q: 6) Using oxirane and bromobenzene, propose a synthesis for the following epoxide: Br 8 (Only sources…
A: It is not necessary during the chemical reaction the reagent is consumed. An organic reaction is a…
Q: [A]. (M) 0.150 0.450 0.450 [B], (M) 0.300 0.300 0.150 Using the information in the table, the value…
A: Answer:Power of concentration term of a reactant in the rate law is the order of reaction with…
Q: find the molar mas and the idenity of M
A: we have given a metal M form a compound with the formula MCl4 and the compound is 74.74% chlorineWe…
Q: Does the molecule below exist as a pair of enantiomers? If so, change the bonds to wedges and dashes…
A: Given that, the structure of the molecule is:
Q: a. How many sigma bonds, including bonds to Hydrogen, are in this compound? b. How many pi bondes…
A: Write the expanded structure of the given compound.If there is a single bond consider it as a sigma…
Q: 20. Which of the following statements is TRUE? A. The total number of molecular orbitals formed…
A: The objective of the question is to predict which of the given statements are true.
Q: What are A, B, and C in the following table, and explain each of them briefly? E₁ A: M hv=E₂-E₁ M B:…
A: In quantum mechanics, electrons in atoms or molecules occupy specific energy levels or orbitals.…
Q: Provide the correct systematic name for the compound shown here.
A: The longest possible continuous chain is selected as parent carbon chain.The functional group is…
Q: to vaporize a 14.9 gram sample of liquid Br2 requires 2.58 kj of heat. what is delta Hvap for Br2
A: To vaporize a 14.9 gram sample of liquid Br2 requires 2.58 kJ of heat. We have to find the value of…
Q: A student combines Fe(NO3)3 solution and KSCN solution to produce a solution in which the initial…
A: The objective of this question is to determine the equilibrium concentration of FeSCN2+ in mol/L.…
Q: Please don't provide handwritten solution ....
A: Check belowExplanation:
Q: The normal boiling point of mercury (Hg) is 356.7 °C. At what temperature (in °C) would the vapor…
A: Given;Initial pressure (P1) = 1 atm (at normal boiling point)Final pressure (P2) = 0.2450 atmInitial…
Q: If we increase the pH (by decreasing the [H+]), the concentration of HCOO (increase/decrease/remain…
A: The question is asking about the effect of changing the pH (or the concentration of H+ ions) on the…
Q: The reaction C(s) + 2 H₂(g) = CH₂(g) has Kp = 0.263 at 1000. K. Calculate the total pressure at…
A: The reaction given is,C(s) + 2H2(g) CH4(g) Kp = 0.263 at 1000K.Part 1Amount of H2 used = 5.135…
Q: Which of the following molecules will not have a 180\deg bond anglea) SO ? b) KrF 2c) SiOd) BeF2e)…
A: The objective of the question is to identify the molecule that does not have a 180-degree bond angle…
Q: 27.22 g of X combines with 84.10 g of Y to form XY. If the molar mass of X is 33.42 g mol–1, what is…
A: The mass of X is 27.22 g.The mass of Y is 84.10 g.The molar mass of X is 33.42 g.mol-1.The formula…
Q: d) e) Z: :Z- =0: :Ö:
A: The index of hydrogen deficiency (IHD) is determined using the following equation:
Q: 7) Which of the following reactions would give the product(s) indicated in substantial amounts…
A: All are F. C. Alkylation reaction Find out good yield pathaway.
Q: 5.4 (+0.2) x 4.6 (+0.3)
A: Here, it can be observed that the multiplication equation is given and needs to determine the…
Q: The rate constant for the decomposition of N O, is 7.78 107 at 273 K and 3.46 10 at T₂. If the…
A: Answer :As per the Arrhenius equationln(K2/K1) = Eact / R (1/T1 - 1/T2)K2= 3.46*10-5K1= rate…
Q: 2. Which mechanism is consistent with the experimental kinetic data for the reaction of the…
A: Answer:Law of mass action states that rate of reaction will be directly proportional to the active…
Q: A 1.13 g sample of an unknown bromide salt is dissolved in water. An excess of AgN 3(aq) is added,…
A: To determine the mass percentage of bromine (Br) in an unknown bromide salt and identify the salt,…
Q: (4) Tell whether the following pair is identical, constitutional isomers, stereoisomers, or…
A: A pair of substances which are non-super imposable mirror images are called enantiomers.A pair of…
Q: What is the oxidation state of carbon in CHF3?
A: The important points to determine the oxidation state of a neutral compound are : The overall…
Q: 2. The normal boiling point of the metal cesium (Cs) is 670.8 °C. At what temperature (in °C) would…
A: The objective of the question is to find the temperature at which the vapor pressure of cesium would…
Q: Give each of the following amines an IUPAC name.
A: The IUPAC Naming of any compound can be given easily by following simple rules - Longest carbon…
Q: The products in the following reaction are incorrect, briefly explain why the products are…
A: Given is organic reaction. The given products are incorrect. The given reaction is,
Q: Y AICI, + HCI + "CN
A: Electrophilic site is a site where lack of electron density. A nucleophilic site is a site where…
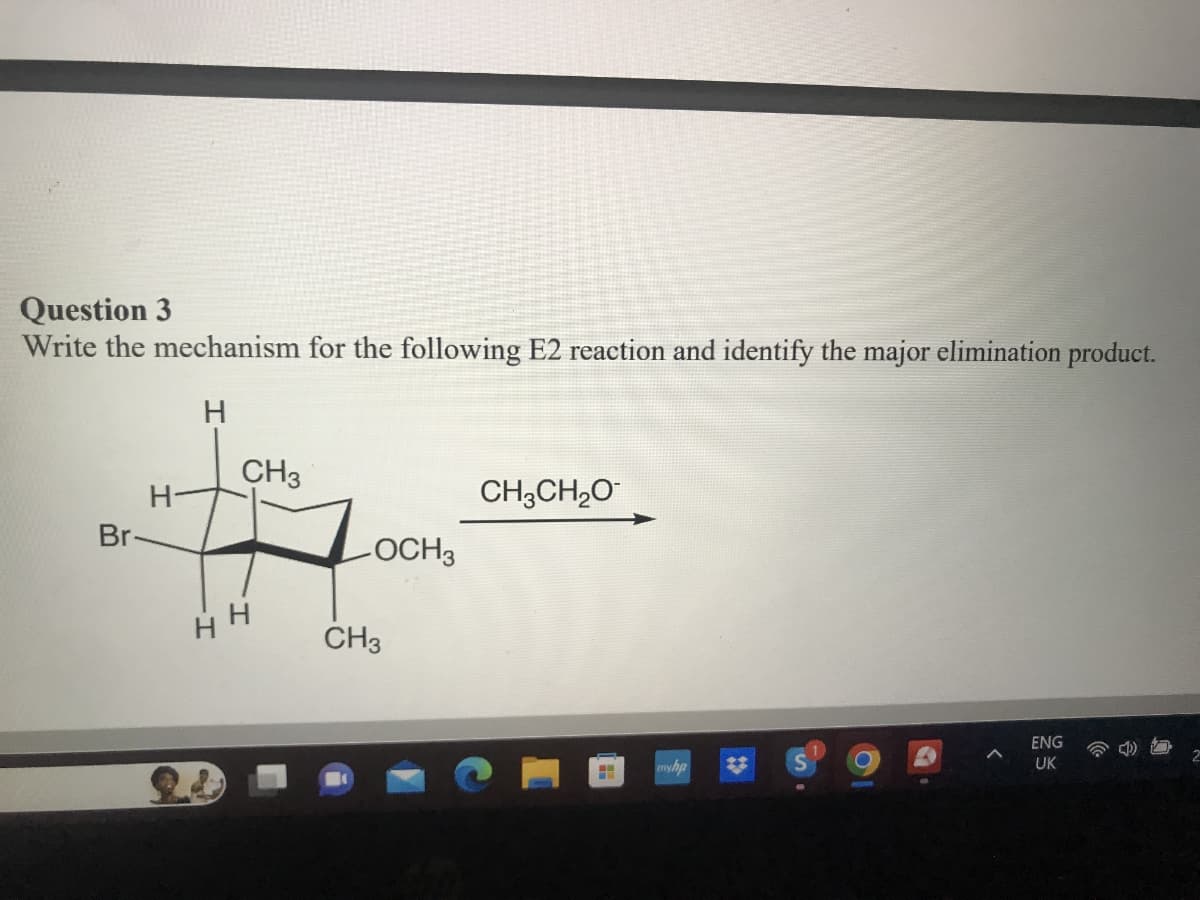
Q: Q4: What is the correct reagent for this reaction: Br major minor
A: Elimination Reaction:Dehydrohalogenation ( Formation of alkenes):Saytzeff's rule : The more highly…
Q: An aqueous solution of 0.045 M HA (pKa = 11.11) is prepared. What is the pH of this solution? Enter…
A: The objective of this question is to calculate the pH of an aqueous solution of a weak acid HA with…
Q: A student combines Fe(NO3)3 solution and KSCN solution to produce a solution in which the initial…
A: The objective of the question is to determine the equilibrium concentration of SCN- in mol/L. This…
Q: When a 28.5 mL sample of a 0.377 M aqueous hydrocyanic acid solution is titrated with a 0.311 M…
A: In first step, we calculate moles of acid and base by multiplying their molarity with volumes. nbase…
Q: Identify the compound(s) that will readily undergo McLafferty rearrangement during a mass spectral…
A: The McLafferty rearrangement is a process observed in mass spectrometry where organic molecules…
Q: Question 4. Which of the following are possible chair conformations of the molecule shown below?…
A: The planar form of a cyclohexane molecule is highly unstable due to high-angle strain. The most…
Q: An unknown compounc has a AH fus of J 20.2k ol and a AS m fus
A: As we know , Entropy of transition (S) = Htrans/ T
Q: Which reagents would be best to achieve the following synthesis? mo OH A) 1) 03 2) Zn, H₂O B) 1) 03…
A: Conversation of Alkene into acidFind out suitable reagent.
Q: EL SU Draw the major product of this reaction. Ignore inorganic byproducts. 1. NaNHz 2.…
A:
Q: When [F] is decreasing at 0.15mol/Lxs, how fast is [G] increasing? Be sure to use correct number of…
A: The rate of any reaction is defined as the change in concentration of reactants to products per unit…
Q: Provide the correct common name for the compound shown here. CH3 H3C H3C -C Br
A: Provide the correct common name for the compound shown here:
Q: Which of the combinations below will produce an insoluble salt? a) Ba(OH)2 + HCI b) MnSO4 + Pb(NO2)2…
A: The slats that do not dissolve in water are insoluble salts. 1) Ba (OH)2 +2HCl ---------> BaCl2…
Step by step
Solved in 3 steps with 3 images

- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.
- Substitutions vs elimination. Pls help me identify the major products and the dominant mechanism. (SN1/SN2/E1/E2 or their combination)Under forcing conditions, sp2 CH bonds can be deprotonated, and sp2 chlorides can be eliminated. Draw two different mechanisms to account for the formation of the isomeric products C3H4 from the same chloride starting material.E 21 write mechanisms for the given reactions
- OCHEM... Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane is heated with methanol. Draw clear structural formulas of all relevant species and use curved arrows to represent electron flow. Also, indicated which step is likely to be rate-determining.Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.(with explanation)
- I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.Explain the possible mechanism (Sn1/Sn2/E1/E2) given the reaction. Show the transfer of electrons/groups, and name the product.I need help to explain the mechanism below in steps in to words Preparation of Cyclohexanone by Hypochlorite Oxidation explain the mechanisms



