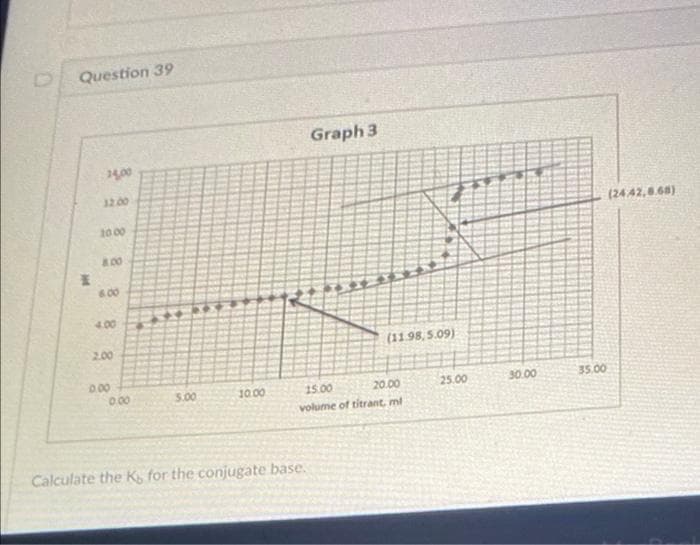
Question 39 pt 14,00 10.00 8.00 600 4.00 2.00 0.00 0.00 3.00 10.00 Graph 3 Calculate the K, for the conjugate base. (11.98,5.09) 15.00 20.00 volume of titrant, ml 25.00 30.00 35.00 (24.42,8.68)
Q: 80. Which one of the following metal oxides does not usually react with sodium hydroxide? (a) Copper…
A: Sodium hydroxide (NaOH) is basic in nature, so it reacts with compounds which are acidic.
Q: 760 mmHg equals 101.325 kPa. What is a pressure of 746 mmHg in kPa? 220.2 kPa 150.3 kPa 84.2 kPa…
A: Given data : 760 mmHg = 101.325 kPa. To calculate : pressure of 746 mmHg in kPa
Q: Spectrum 6 LOD TRANSMITTANCE D 4000 3000 Important Peaks: Wavenumbers: Name of compound: 2000…
A:
Q: How much heat is required to raise the temperature of 85.0 grams of water by 15.0 degrees C?…
A:
Q: A certain liquid X has a normal freezing point of -5.00 °C and a freezing point depression constant…
A:
Q: For the following "possible" reaction, which are the states of the products in order from left to…
A:
Q: Explain the role of timescale of the experiment and the type of information you get from NMR.
A: role of timescale of the experiment. information what we get from NMR.
Q: Write the expected product for each of the following reaction: 2-Chloro-1-cyclohexanone + MCPBA…
A: We have to write the expected product for each of the given reactions. Note: According to Q&A…
Q: Which of the following salts give acidic aqueous solutions? (choose all that apply) a. NH4NO3 b.…
A: Acid : According to Arrhenius theory, acid are those substance which gives [ H+] in aqueous…
Q: Consider the following system at equilibrium. (Charges have been omitted from the equation) Co(CI)4…
A: Given the temperature of observation is 85 oC
Q: A student produced ZnI2 then allowed the product to cool to room temperature without covering the…
A:
Q: What is the expected M/Z of the base peak of the following compound?
A: The base peak of the following compound will be 57.
Q: For the diprotic weak acid H₂A, K₁1 = 3.8 x 10-6 and K₁₂ = 7.7 x 10-⁹. What is the pH of a 0.0750 M…
A: Given, The concentration of H2A = 0.0750 M. Ka1 = 3.8 × 10-6 and Ka2 = 7.7 × 10-9 The pH of the…
Q: 34. Predict the major organic product of each reaction or provide the reagents needed to complete…
A: I request you to please repost your remaining questions seperately.
Q: Use the IUPAC system to name the structural diagram shown above. О Н нн нн Н-С-С- Н- C - C - 0 Н Н н…
A:
Q: Write the subshell electron configuration (i.e.1s² 2s², etc.) for the Fe26 atom.
A:
Q: the reference sheets for all equilibrium constant values needed. 1. Which of the following salts has…
A:
Q: Ethanethiol, C₂H5SH, is an odourous compound added to natural gas so that leaks can be detected. It…
A:
Q: A 195 mL sample of ocean water contains 7.0 gg of NaCl. What is the molarity of the solution with…
A: Given, Mass of NaCl in ocean water= 7.0g Volume of ocean water = 195 mL Molarity of NaCl solution…
Q: Br Н НО H+ ОН ин 1. PPh3 2. BuLi 1. 2. H2O, H+ Н (0)
A: The Wittig reaction is a reaction of an aldehyde or ketone with a triphenyl phosphonium ylide…
Q: Which of the following statements is false? Unlike the volume of a solid or a liquid, the volume of…
A: FALSE statement is :
Q: What will be the resulting organic products’ structure when the following reactions occur and…
A: When (R)-2-iodobutane show SN2 mechanism when reacts with NaOCH3 as shown below:
Q: pectrum 7 OD 50 D 4000 3000 mportant Peaks: Wavenumbers:_ ame of compound: 2000 HAVENUMBERI-I way…
A:
Q: Nickel has a face-centered cubic unit cell with a length of 352.4 pm along an edge. Calculate the…
A: Given, Nickel has a face centred cubic unit cell. Note: 1 pm = 10-10 cm Edge length (a) = 352.4 pm =…
Q: What will be the resulting products structure when p-methylphenol reacts with HNO3
A:
Q: At 280 K, a gas has a volume of 1.5 L, and a pressure of 1.0 atm. The gas is heated to 380 K, and…
A:
Q: Question 2: The molecular formula of acetylsalicylic acid (aspirin), one of the most commonly used…
A: Dear student since you have posted multiple questions we will solve first question for you if you…
Q: Provide the missing reactants, products or reagents for the following reactions. When more than one…
A: NaBH4 is a mild reducing agent . It converts aldehyde, ketone into alcohols.
Q: Determine the equilibrium concentration of H2O(g) for the following reaction with the equilibrium…
A:
Q: Nitrogen gas reacts with hydrogen gas to produce ammonia via the following reaction: Part A If you…
A:
Q: Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers,…
A: Stereoisomer are those which have same molecular formula but have different orientation in 3D space.…
Q: 8. Propose two feasible syntheses of the amine shown below, starting from benzene and any needed In…
A:
Q: Select all that shifts equilibrium right. CO(g) + Cl₂(g) → COCI₂(g) AH = -107.6 KJ A) More Cl₂ is…
A: Le Chatelier's principle: According to the Le Chatelier's principle when a change in concentration…
Q: The equilibrium constant (K) for the reaction PC13 (g) + Cl₂(g) → PCls (g) equals 49 at 230°C. If…
A: According to the question we have a chemical reaction PCl3 g + Cl2 g ↔ PCl5g The equilibrium…
Q: Criven ka = 2.5×10-9 Find the pH at a 0.724 M Savetion of the weak Jose bese Bro- ?
A:
Q: Question 3: A simple triglyceride is one that has all three fatty acid chains identical to one…
A: According to guidelines we are supposed to answer only one question
Q: Give the IUPAC name for the following structures: (i). (iii). (ii). (iv).
A: Given compounds are : Give IUPAC names of these compounds = ? If more than one double bonds are…
Q: Hello. Thank you for answering my question. I actually tried 92.5 kJ/mol-1 and also 92.484 kJ/mol…
A:
Q: 7 lity " Cl, Br, I Halides ity Most / Sr+2 / Sn+4 =/Pb+2/Mg¹2 Ag*/Pb+2 /Ba+2/Pb+2 Solubility Table…
A: We can do quantitative analysis to separate the cations as well as anions in solution mixture.
Q: What molecules have trigonal planar ELECTRON PAIR GEOMETRY A) O3 B) HCl C) ClO3- D)NaCl E)CH2O…
A: -> For trigonal planar geometry there must be three electron pair. Electron pair = bond pair +…
Q: Draw the five isomeric alkanes of molecular formula C6H14. Determine how many proton environments…
A: Since, Isomers are those whose molecular formula is same but connectivity of substituent is…
Q: 11-7 What is the minimum mass of ethylene glycol (C₂H5O₂) that must be dissolved in 14.5 kg of water…
A: Here we are required to find the mass of ethylene glycol must be dissolved to have freezing point…
Q: A quantity of helium gas is originally held under a pressure of 145 kPa at 18°C. If the volume of…
A:
Q: The standard AGsys for a neutralisation reaction is exergonic by 8,1kcalmol-¹. This would correspond…
A:
Q: 3. A buffer sol'n is prepared by dissolung Potassium fluoride in hydroféburic acid. J Suppose you…
A: Given, A 500.0 mL of buffer solution contain, Molarity of KF = 0.13 M Molarity of HF = 015 M The…
Q: Explain the main differences between flame atomic absorption and UV-VIS molecular absorption in…
A:
Q: A 50.0 mL solution of Ca(OH)₂ with an unknown concentration was titrated with 0.340 M HNO3. To reach…
A:
Q: Predict the major product for the following reaction u OH A) on H₂O* pyridine Br₂ Dy Br
A:
Q: Identify the catalyst in the following reaction mechanism steps. Step 1:…
A: Catalyst : A catalyst is a chemical species that enhances the rate of a chemical reaction by…
Q: How would you describe the molecular geometry of C2H2F2, and what are its bond angles?
A:
Step by step
Solved in 5 steps with 1 images

- An aqueous solution containing 0.16 M of unknown base has a measured pH equal to 7.51. What is the value of pKb for the unknown base? Include 3 significant figures in the answerAlanine is a diprotic amino acid with a pKa = 2.344 for the carboxylic group and a pKa = 9.868 for the ammonium group. Estimate the pH of a solution of 0.340 M alanine. Answer using two significant figures.Ajja what is the ph of a 0.10m solution of an acid with a pKa of 8.4?
- A 50.00 (±0.02) mL portion of an HCl solution required 29.71(±0.02) mL of 0.01963(±0.0032) M Ba(OH)2 to reach an end point with bromocresol green indicator. ? of HCL = 29.71?? ? 0.01963 ???? ??(??)2 ?? ? 2 ???? ??? ???? ??(??)2/ 50.00?? = 0.02333 ? Calculate the uncertainty of the result (absolute error).Calculate the coefficient of variation for the result.What is the Ksp expression for PbCl2 in water? Group of answer choices Ksp = [Pb2+][Cl–]2 Ksp = [Pb2+][Cl–] Ksp = [??2+][??−]2[????2][Pb2+][Cl−]2[PbCl2] Ksp = [??2+][??−][????2][?2?][Pb2+][Cl−][PbCl2][H2O]15. A 300.00 mL solution of 0.00165 M A2B5 is added to a 230.00 mL solution of 0.00380 M C2D3. What is pQsp for A2D5?
- ANSWER THE FOLLOWING WITH A COMPLETE SOLUTION. Compute for the pKa of benzoic acid (Ka = 6.3 × 10-5). What is the Kb of ammonia if its pKb is 4.75? What is the [OH-] of a solution if its pH is 5.69? Identify the [H+] of a solution with a pOH of 11.41. Identify if it is neutral, acidic or basic. What is the pH of a buffer prepared with 0.51 M disodium phosphate and 1.1 M sodium acid phosphate? (pKa of sodium acid phosphate = 7.21) Compute the pH of a buffer containing 0.62 M boric acid (Ka = 5.90 × 10-10) and 0.45 M sodium borate.In the spectrofluorometric analysis of quinine, which of the following statements/phrases can be considered a factor level? A. concentration of 2ppm of iodide B. concentration of quenchers C. use of quenchers D. use of iodidePhenolphthalein has a pKa of 9.7 and is colorless in its acid form and pink in its basic form. For pH= 7.7 calculate [In−]/[HIn]. Express your answer using two significant figures.
- Nitrous acid has a Ka of 4.5×10-4. a.) What is the pH of a buffer solution containing 0.13 M HNO2 and 0.15 M NO2- ? Express your answer using two decimal places.The value of Ksp for Mg3(AsO4)2 is 2.1 x 10-20. TheAsO43 - ion is derived from the weak acid H3AsO4 (pKa1 =2.22; pKa2 = 6.98; pKa3 = 11.50). (a) Calculate the molarsolubility of Mg3(AsO4)2 in water. (b) Calculate the pH of asaturated solution of Mg3(AsO4)2 in water.Data Table 2: Complexometric Titration of Tap Water photo shows equation to find water hardness 6 ml of water used (5 ml of tap water 1ml of buffer) Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading (mL) 2ml 2ml 2ml Final Syringe Reading (mL) .61ml .69ml .58ml Volume of EDTA Consumed (mL) 139ml 131ml 142ml Water Hardness ppm CaCO3 (mg/L) Average ppm CaCO3 (mg/L):

