Q: Please provide the name of the dienes and dienophiles, and their products and explain the reaction…
A: The names of diene and dienophile for the formation of the given compound iDials-Alder reaction. The…
Q: 3. SiI4 - use a black ball for silicon when constructing this model Total # of Valence Electrons: 32…
A: sp hybridization -----> Linear sp3 hybridization -----> Tetrahedral
Q: Arrange the following in terms of decreasing acidity. Type the letter labels for each structure on…
A: Acidic strength of organic acids like carboxylic acids, alcohols and phenols depends upon its…
Q: 7 O+Y 8 ¿+Ngi ← OSŤ 150 8 +0g + H+¿
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: Question 21 ( Show the preparation of phenylacetic anhydride starting from 2-phenylethenol
A:
Q: Choose four compounds that are most soluble in water.
A: Given:
Q: As explained in the book, the Voltaic cell is where the two components of a spontaneous redox…
A: In a Galvanic cell, Zn(s) undergoes oxidation to Zn2+(aq) and Cu2+(aq) undergoes reduction to form…
Q: small.unt of sodium chloride (NaCl) is dissolved in a large amount of water. Imagine separating this…
A:
Q: A student conducts a titration using NaOH (strong base) as the titrant to determine the…
A:
Q: 28 What is the accepted IUPAC name for the following hydrocarbon? a Place your answer in the box and…
A: Ans. Given hydrocarbon have 7 carbon longest chain and propyl group is at 4 no. Carbon 7 Carbon -…
Q: ▪ In a blood alcohol analysis by GC-FID, methanol elutes at 1 min, ethanol at 2 min, IPA at 3 min,…
A: Answer : Relative retention time = retention time of analyte / retention of known peak based on…
Q: mL buffer solution is 0.250 M in acetic acid and 0.250 M in sodium acetate. (a) What is the initial…
A: Given→ Volume of buffer solution = 250.0 mL Molarity of acetic acid = 0.250 M…
Q: QUESTION 3 a) Propose a synthesis route to prepare 3-ethylpentan-3-ol from 1-bromopropane. Note that…
A: 3-ethylpentan-3-ol is tertiary alcohol. Tertiary alcohols can be prepared from Grignard reagents…
Q: For the Lewis dot structure shown below, predict: Y=X=Y (b) what is the bond angle?
A:
Q: QUESTION 2 a) Determine the degree of unsaturation of the following compounds: i. C15H30N16Br10…
A: Since you have asked multiple question, as per our company guidelines we are supposed to answer…
Q: Synthesis #2 CHPh H from compounds with 6 carbons or fewer
A: To get the reactant molecule for synthesizing this molecule we have to carry out Retro-synthetic…
Q: A 1.132 g sample of KHP (HKC8H404) required 28.05 mL of NaOH solution to reach endpoint. Calculate…
A:
Q: True or false: 2,2-dimethylpropane is a structural isomer of n-pentane
A: Given statement: 2,2-dimethylpropane is a structural isomer of n-pentane. We have to find whether…
Q: An unknown compound has the following spectroscopic properties: Mass Spectrometry: m/z 102 (very…
A: 1H-NMR is the main technique used for the identification of complete structure of the compound other…
Q: A sample of O2 gas was collected over water at a total pressure of 620 mm Hg and 25 degrees c. The…
A: Dalton's law: Dalton's law of partial pressures states that in a mixture of non-reacting gases, the…
Q: In titrating a H2SO4 solution, it was found that 23.66 mL of 0.2137 M NaOH would neutralize 22.04 mL…
A:
Q: What is the correct IUPAC name of the following compound? H a Problem viewing the image. Click…
A: Here we have to write the IUPAC name of the following given compound . * Longest continuous carbon…
Q: AATT You are a researcher in a geochemistry laboratory. A colleague brought to you a new mineral…
A: Here we are required to find the mass percent.
Q: In SN2 mechanism, the intermediate produced is a carbocation. True False
A:
Q: [4.05819 Mass of Urea (g): Buret Initial Volume (mL): water Part 2 Calculation: Final Temperature of…
A:
Q: Using the stock solution concentrations of 0.20 M Fe3+ and 2.0*10^-3 M SCN-, calculate the following…
A: Stock solution concentrations of 0.20 M Fe3+ and 2.0*10-3 M SCN- 2ml of Fe3+ and 0.25 ml SCN-…
Q: Select all of the reactions that will provide a product as a single enantiomer. 0 Br Br Br !!!!!H…
A: Here we have to predict the reactions which will produce only a single enatiomer as a major product.…
Q: How many moles of Na2CO3 are there in 10.0 L of 2.00 M solution? Show a clear solution.
A:
Q: A 0.600 g sample of steel is dissolved in acid, and the manganese is oxidized to permanganate. The…
A: Given → Mass of the sample of steel = 0.600 g Volume of solution = 100 mL Wavelength = 520 nm Length…
Q: 5. For the chemical reaction below (please note, this is the same as CH4 + Br2 → CH3Br + HBr; the…
A: when there is sufficient difference in electronegativity , then that bond is polar bond where if…
Q: 38) A reaction is found to have an activation energy of 108 kJ/mol. If the rate constant for this…
A:
Q: Draw the structures of the compounds below and give the number of peaks observed in their 1H and 13C…
A:
Q: Arrange the following carbonyl compounds in INCREASING REACTIVITY towards nucleophilic addition.
A:
Q: A ¹H NMR spectrum is shown for a molecule with the molecular formula of C6H15N. Draw the structure…
A: using the NMR spectra and the molecular formula of the compound, we have to predict it's structure
Q: What is the major organic product of the following Claisen Condensation reaction? excess OMe 1. Na…
A: Question belongs to mixed condensation reaction. Condensation reaction between aldehyde and ester.
Q: ▪ In a THC metabolite confirmation by GCMS, the metabolite standard curve has a slope of 5ng/ml. In…
A:
Q: C. Arrange the following compounds in order of decreasing reactivity towards hydrolysis. H3CO i ΝΑΙ…
A: Rate of hydrolysis depends on the electrophilic character of carbonyl group. Greater is the…
Q: True or False: Mass spectra are like a fingerprint. Every compound has a unique one. Which mode of…
A: Mass spectra are like fingerprint.every compound has unique one. True because it is different for…
Q: What is/are the reagent(s) and conditions for the following reaction? Problem viewing the image.…
A:
Q: Distinguish between High Density Polyethylene (HDPE), Low Density Polyethylene (LDPE) and Linear Low…
A: High density polyethylene:- a) high density b) stronger intermolecular forces , stronger than low…
Q: 7. Show how the following carbohydrate forms a cyclic structure (hemiacetal). Ho H H (OH HITCH -041…
A:
Q: Which statement is false? 4 electron groups matches with a tetrahedral electron pair geometry For an…
A: Ans. Option (C) is false. Resonance structure is a method of describing de-localized electrons…
Q: На HO A) (В) C) -R 11!!!! нужн 1888
A:
Q: How can controlled potential coulometry detect the analyte?
A: Controlled potential coulometry, as the name suggests, is a coulometry method that measures the…
Q: O 40 50 Transmittance [%] 60 70 80 90 100
A: Answer. First we will find the major peaks from the given IR spectra. 2987 cm-1 medium sharp (alkane…
Q: I am supposed to calculate Average absolute deviation (%). I am not sure if 7.16 is the final…
A:
Q: What is the major organic product of the following reaction? (Assuming excess reagents) A. B. C.…
A: Organic reaction are those in which organic reactant react to form organic products.
Q: 5. NO₂ Total # of Valence Electrons: Lewis Structure (show all resonance structures if applicable)…
A: We have to draw the Lewis structures for the given two molecules and fill the given tables
Q: 16). What is the main product of the following reaction? + Br₂ FeBr3 A) Br B) Br E) No product; the…
A:
Q: 2-Carboethoxycyclohexanone (A) can be prepared by condensation reactions as follows. Path 1: Via…
A: Since you have posted a question with multiple sub-parts ,we will solve first three sub-parts for…
Step by step
Solved in 2 steps with 2 images

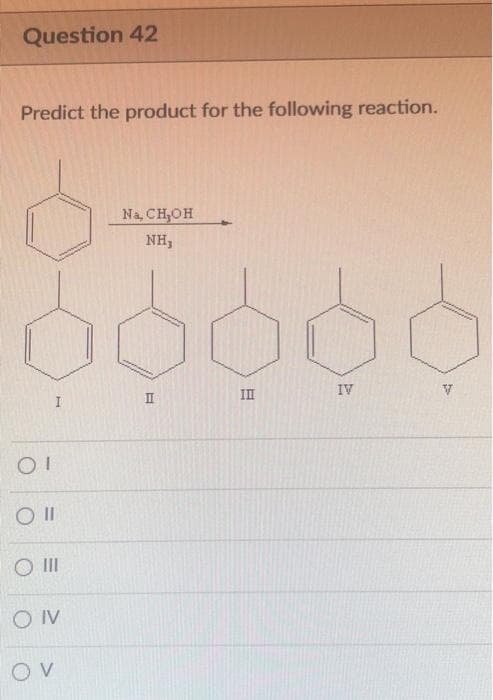
- Draw themajor product of this reaction. Ignore inorganic byproducts. PPC, CH2Cl2QUESTION 4Predict the products(s) from the reaction of 1-hexyne with each of the following reagents:a) 1 mole of HBrb) H2O, H2SO4, HgSO4c) 2 moles of Br2d) Excess Cl2e) 2 moles of HCl with H2O2f) 1 mole of HClPredict the product for the reaction shown
- Do not give handwriting solution. Please predict the product from the following reaction with the four choices listed below it.(please answer all questions) 1) Predict the products from reaction of 1-hexyne with thesereagents: a) One equivalent HBr b) One equivalent Cl2 c) H2 , Lindar Catalyst d) NaNH2 in NH3 , then CH3BrPredict (by name) the major product for the following reaction when NBS/peroxide is added. The starting compound is byblo[4.4.0]dec-1-ene. Note the numbering and name the product.
- Please complete the reactions given below.I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.What is the common name of the product of below

