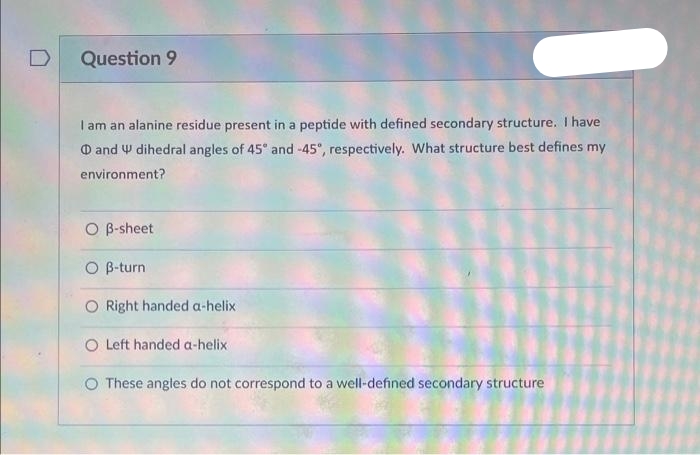
Question 9 I am an alanine residue present in a peptide with defined secondary structure. I have and dihedral angles of 45° and -45°, respectively. What structure best defines my environment? O B-sheet O B-turn O Right handed a-helix O Left handed a-helix O These angles do not correspond to a well-defined secondary structure
Question 9 I am an alanine residue present in a peptide with defined secondary structure. I have and dihedral angles of 45° and -45°, respectively. What structure best defines my environment? O B-sheet O B-turn O Right handed a-helix O Left handed a-helix O These angles do not correspond to a well-defined secondary structure
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter3: Biological Macromolecules
Section: Chapter Questions
Problem 23CTQ: Amino acids have the generic structure seen below, where R represents different carbon-based side...
Related questions
Question
Transcribed Image Text:D
Question 9
I am an alanine residue present in a peptide with defined secondary structure. I have
and dihedral angles of 45° and -45°, respectively. What structure best defines my
environment?
O B-sheet
O B-turn
Right handed a-helix
O Left handed a-helix
O These angles do not correspond to a well-defined secondary structure
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning