* Question Completion Status: QUESTION 14 In an experiment, a student obtained the following masses for a product: 0.651 g. 0.643 g. 0.648 g, 0.609 g, and 0.646 g. What is the average deviation from the mean? O 0.6394 g O 3.12 g O 0.0608 g O 0.012 g
* Question Completion Status: QUESTION 14 In an experiment, a student obtained the following masses for a product: 0.651 g. 0.643 g. 0.648 g, 0.609 g, and 0.646 g. What is the average deviation from the mean? O 0.6394 g O 3.12 g O 0.0608 g O 0.012 g
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 6ALQ: You have a chemical in a sealed glass container filled with air. The setup is sitting on a balance...
Related questions
Question
Transcribed Image Text:Question Completion Status:
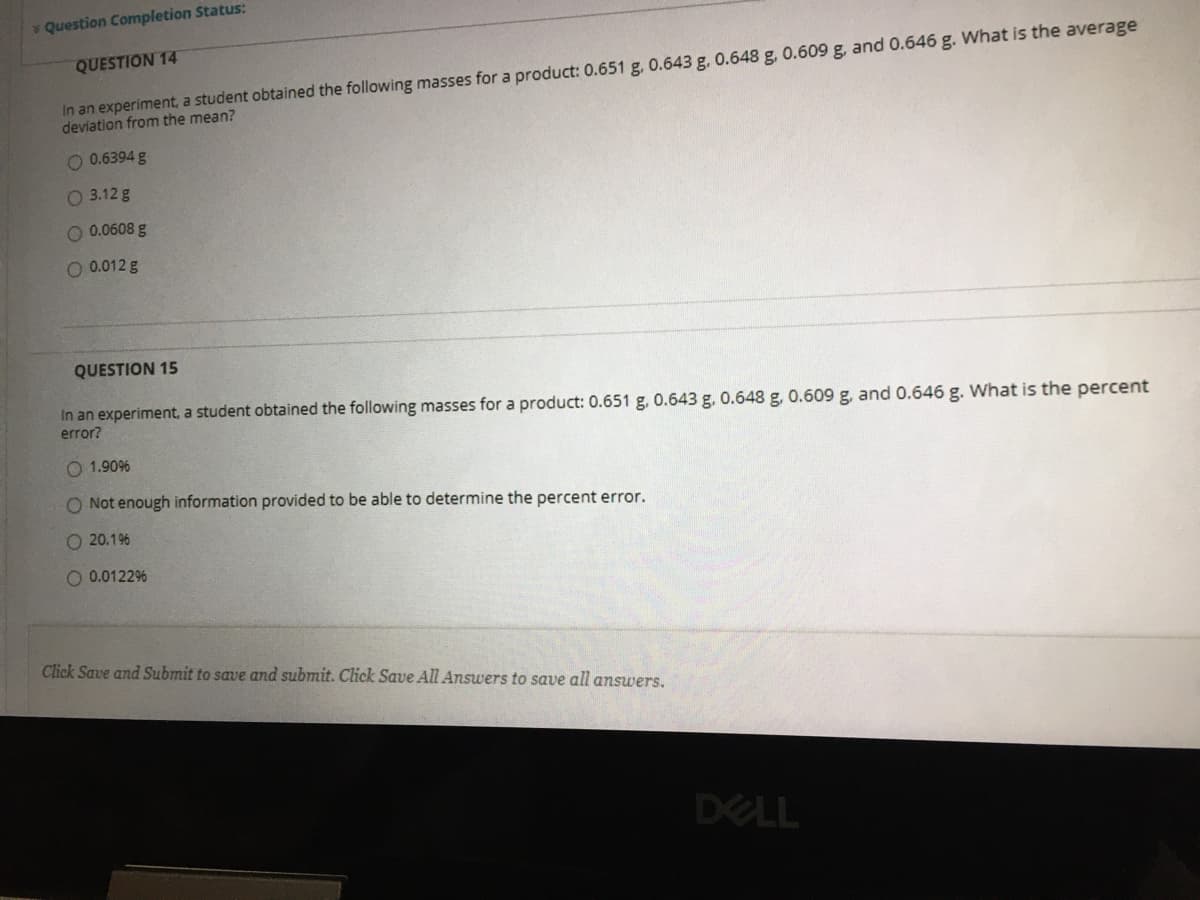
QUESTION 14
In an experiment, a student obtained the following masses for a product: 0.651 g, 0.643 g. 0.648 g, 0.609 g, and 0.646 g. What is the average
deviation from the mean?
O 0.6394 g
O 3.12 g
O 0.0608 g
O 0.012 g
QUESTION 15
In an experiment, a student obtained the following masses for a product: 0.651 g, 0.643 g. 0.648 g, 0.609 g, and 0.646 g. What is the percent
error?
O 1.90%
O Not enough information provided to be able to determine the percent error.
O 20.196
O 0.0122%
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
DELL
Transcribed Image Text:Question Completion Status:
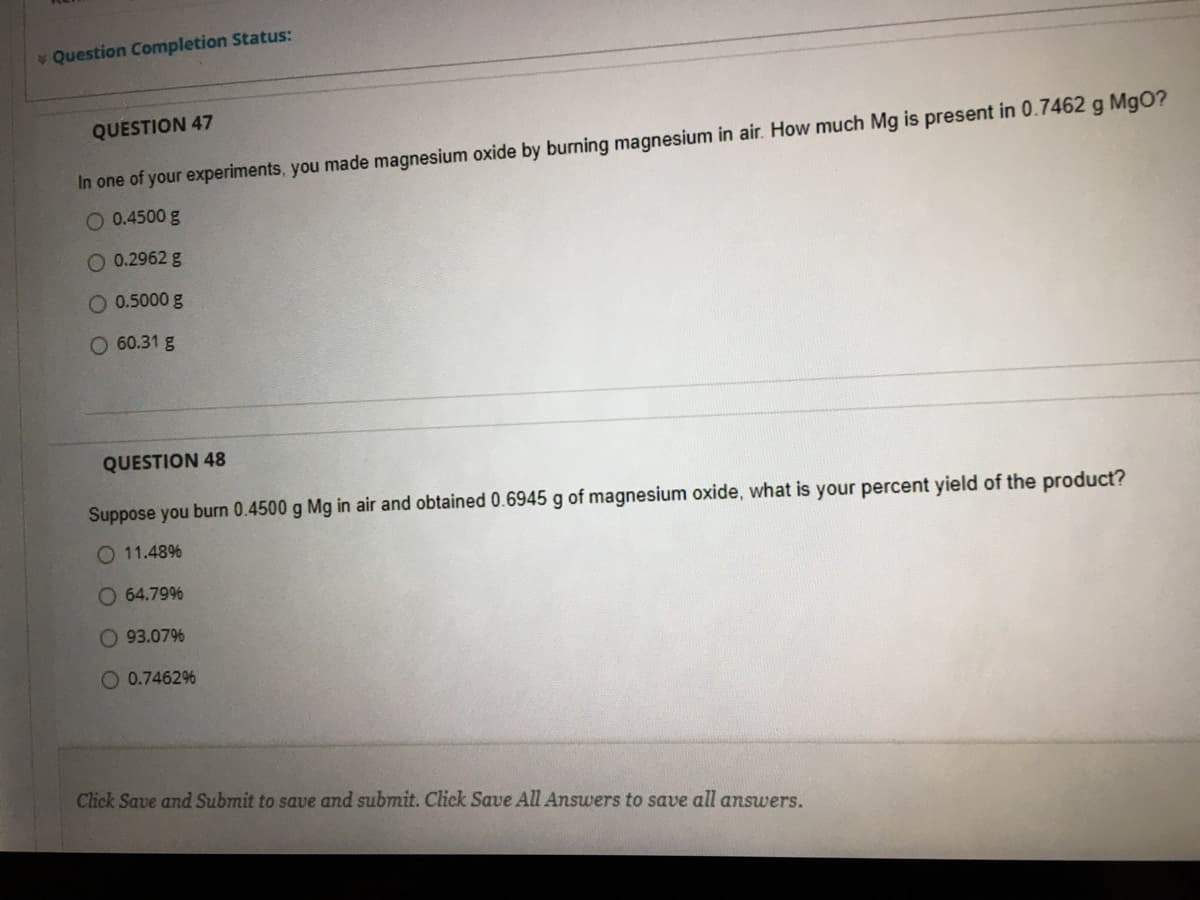
QUESTION 47
In one of your experiments, you made magnesium oxide by burning magnesium in air. How much Mg is present in 0.7462 g MgO?
O 0.4500 g
O 0.2962 g
O 0.5000 g
O 60.31 g
QUESTION 48
Suppose you burn 0.4500 g Mg in air and obtained 0.6945 g of magnesium oxide, what is your percent yield of the product?
O 11.48%
O 64.79%
O 93.07%
O 0.7462%
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning