* Question Completion Status: QUESTION 4 Roaches for sale | C... For Sale/Trade/Wan... * Tarantulas CSL Behring MHell Let Loose TR St... Indicate which three of the following statements about the electron in a hydrogen atom are true by checking the box in front of each true statement. The atom is in the ground state when the electron is in the n=1 level. O Energy is released when the electron is excited from the n=1 level to the n=3 level. A shorter wavelength of light is emitted when the electron moves from the n-3 level to the n=2 level than when the electron moves from the n=4 level to the n=2 level. O Energy is always released when an electron relaxes. A lower frequency of light is emitted when the electron moves from the n=3 level to the n=2 level than when the electron moves from the n=4 level to the n=2 level. The atom is stable when the electron relaxes to the n=2 level.
* Question Completion Status: QUESTION 4 Roaches for sale | C... For Sale/Trade/Wan... * Tarantulas CSL Behring MHell Let Loose TR St... Indicate which three of the following statements about the electron in a hydrogen atom are true by checking the box in front of each true statement. The atom is in the ground state when the electron is in the n=1 level. O Energy is released when the electron is excited from the n=1 level to the n=3 level. A shorter wavelength of light is emitted when the electron moves from the n-3 level to the n=2 level than when the electron moves from the n=4 level to the n=2 level. O Energy is always released when an electron relaxes. A lower frequency of light is emitted when the electron moves from the n=3 level to the n=2 level than when the electron moves from the n=4 level to the n=2 level. The atom is stable when the electron relaxes to the n=2 level.
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter7: Quantum Theory Of The Atom
Section: Chapter Questions
Problem 7.24QP: Investigating Energy Levels Consider the hypothetical atom X that has one electron like the H atom...
Related questions
Question
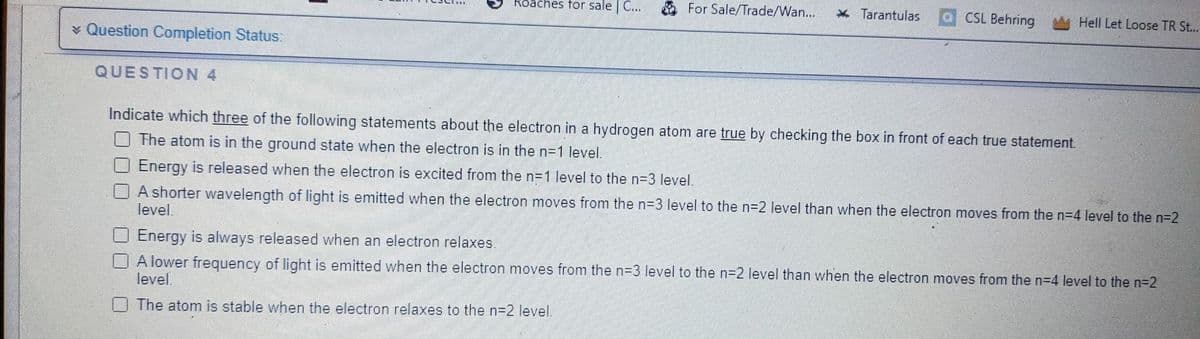
Transcribed Image Text:> Question Completion Status:
QUESTION 4
Roaches for sale | C...
For Sale/Trade/Wan... * Tarantulas
CSL Behring
Hell Let Loose TR St...
Indicate which three of the following statements about the electron in a hydrogen atom are true by checking the box in front of each true statement.
The atom is in the ground state when the electron is in the n=1 level.
Energy is released when the electron is excited from the n=1 level to the n=3 level.
A shorter wavelength of light is emitted when the electron moves from the n=3 level to the n=2 level than when the electron moves from the n=4 level to the n=2
level.
Energy is always released when an electron relaxes.
A lower frequency of light is emitted when the electron moves from the n=3 level to the n=2 level than when the electron moves from the n=4 level to the n=2
level.
The atom is stable when the electron relaxes to the n=2 level.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning