Q: Which hydrogen in each molecule is more acidic? b. NH3 or H2O c. HBr or HCl d. H2S or H2O
A:
Q: h) Which is a weaker base and why? Pyridine Pyrrole i) Name the following compounds. H2N A B NH2
A: Huckel's rule for aromaticity: The compound must be cyclic planar geometry with (4n+2) pi electrons.…
Q: H2 NH2 NH3 -NH2 -NH2 с. O,N CH d. CgH;NH, (CHs)2NH -NH2
A: In case of nitrogenous compound greater the availability of lone pair on nitrogen atom greater it's…
Q: Which of the following is most weakly basic? O CH3CO2 О но- O CH3CH2O¯ O CH3O"
A: A substance which accepts a proton is known as base. The substance which is formed after the…
Q: 2 Which of the following ranks the compounds in order of increasing basicity, putting the least…
A: Those compound which donate lone pair easily have more basic character
Q: Which compound in each pair is the stronger acid? (See the Attachment)
A: To find out the stronger acid we can look at the pKa value or the Ka , pKa value is one method…
Q: Which compound in each pair is more acidic? H. Н. (а) or I II H Н. (b) or I II Н. (с) or II
A: After removal of H+ if anion is stable then molecule is more acidic and if molecule is unstable then…
Q: Which of the following is the strongest base? a. Cl-CH2CH2NH2 Ob. Cl-CH2CH2CH2NH2 Oc. Cl-aromatic…
A:
Q: 1.(a) Which of the following groups has the LOWEST IUPAC priority? (A) CH3 (B) NH2 (C) OH (D) COOH…
A:
Q: Which compound in each pair is the stronger base? (CH,CH2),NH b. (CH;CH2),NH (CICH,CH2)2NH a. or or
A: Basicity depends on how much the lone pair on N is available for donation to H+ Anything that…
Q: HA Hb CH2=CH-CH-CH=CH2 0r or A
A: Two compounds are given to us:
Q: 5. Rank each of the five compounds by relative basicity. Use a "1" for the strongest base, a "2" for…
A: Given compounds: We have to rank each of these compounds by relative basicity.
Q: (4) Answer both sections () and (1). O Arrange the following compounds in order of increasing boling…
A: The given questions are detailly answered below,
Q: Rank the compounds in each group in order of increasing basicity.
A:
Q: Rank the following compounds in order of decreasing acidity. List the most acidic first. OCH3 `NHCH3…
A: In a simple definition, the compound which loses the proton H+ then it is said to be acidic. If the…
Q: Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used…
A: Since the basicity of N depends on 2 factors. 1) whether its lone pair electrons are involved in…
Q: Which compound would be the weakest acid? O CH3CHCICHCICO2H O CHCI2CH2CH2CO2H CH3CCI2CH2CO2H O…
A: minus Inductive effect is directly proportional to acidic character
Q: 4. For each of the following acid/base equations: a. draw the products, b. mention whether the…
A: Acid base reaction: An acid–base reaction is a chemical reaction in which an exchange of one or more…
Q: 1. Which nitrogen-containing compound of each pair would you expect to be stronger base? Indicate…
A:
Q: Rank the compounds in each group in order of increasing basicity.
A: Basicity- The ability of compound to donate lone pairs of electrons or accept protons is called…
Q: i. (a) (b) O,N- -NH, NH2 or O,N- ii. (a) H;C (b) CH3 CH3 H;C N H3C, „CH3 or
A:
Q: 2. Which of the following compounds is the strongest acid? B. CICH2COOH C. CH3CH2OH A. CH3COOH D.…
A:
Q: Rank the compounds in each group in order of increasing acidity.
A: a. When we see the first set of compound given we compare the electronegativity of the halogens ,…
Q: Rank the following compounds in order of increasing basicity: NH₂ NH₂ NH₂ NH₂ A) B) D) OCH3 NO₂ 2
A:
Q: Which of the following compounds is most basic? CH4 NH3 H2O HF
A: We know that conjugate of most acidic compound is weakest base
Q: Which of the following compounds is the strongest base? O CH3CH2CH2MgBr O CH3CH2CO2MgBr O…
A: A base is a substance that donates electron.
Q: Which of the following anions is most basic? A) H3C: B) C) H¿C=CH D) HC=C:O В
A: Strength of base or basicity of anion depends upon the conjugate base of the acid. Greater the…
Q: Q.14 Arrange the following compounds in order of increasing basicity. Aay CH;NH, CH, NH , CH,NH- b…
A:
Q: 19. Which of the following species is the strongest base? А) НО В) H2N C) CH3CОO D) CI 20. Which of…
A: The conjugate of weak acid is strong base Option B is correct , that is amide Ion is most basic…
Q: Somew 2. Consider the following compounds. NH
A: The compounds given are conjugate base of CH3COOH and CH3CONH2. So given anions are base.
Q: Rank the labeled protons in each compound in order of increasing acidity.
A: 1) the order of acidity for the mentioned H will be b > a > c This is because when we remove…
Q: CH,NH, -2 NH2 CH3 or (D) (C)
A: According to Bronsted Lowry theory atoms/molecules/ions which accept protonH+ are known as bases.…
Q: А. H CH3 CH3 HO-S-CH3 + H-0-CH3 .. B.
A:
Q: 3. For each pair of compounds, circle the strongest base: b. VS. Vs. a. OH NH2 HO HS d. vs. vs.
A: Since you have posted question with multiple subparts , as per guidelines we can solve ist three. If…
Q: Which Nitrogen containing compound is the most basic? B None of these OC (A) A H-CEN (B) NH4 (C) N.…
A: Lone pairs which are easily available for donation of electrons are considered as basic in nature.…
Q: Which compound is stronger base in each set of compounds? (ii) -CH,NH; a. (i) (üi) -NH2 CONH, b. (i)…
A: Ammonia is less basic than the primary, secondary, and tertiary alkyl amines because of the…
Q: Which of the following acids is the strongest acid? a) FCH2CO2H b) CH3CH2OH c) CH3CO2H d)…
A: Acidic strength depends on the stability of conjugate base. After removal of proton, conjugate base…
Q: СООН ОН CH3 СООН NO2 CI
A: The order of acidity is as follows: I>II>IV>III Acidity is basically, the tendency to loose…
Q: Identify the more acidic compound from the structures below: NH2
A: Explain the acidity order in the given structure-
Q: Rank the following alcohols from strongest to weakest acid. b. Explain the relative acidities. CH2…
A: a. Analyzing the given compounds shows that 3 different types of functional groups are attached to…
Q: Rank the following in order of increasing basicity. The first compound in your list should be the…
A: The basicity of the compound depends on the availability of the lone pair of electrons. More is the…
Q: 1. Identify which of the labeled hydrogens in the following compounds is the most acidic. H-…
A: Acids are those substance which gives H+ ion on dissociation. The compound which gives H+ ion either…
Q: Rank the following compounds from strongest to weakest acid: CH3CH2OH CH3CH2NH2 CH3CH2SH CH3CH2CH3
A: Ethanethiol is more acidic than ethanol because of the large size of sulfur atom as compared to…
Q: 5. Explain which of the following pairs would be the stronger acid and why a. CH3CH₂CHCICOOH or…
A: In question 5, we have to select the following pair between a and b option be stronger acid and In…
Q: Rank the following compounds in order of increasing basicity. CH3 H3C° CH3 II 4® // H3C- CH3 CH3 N.…
A: Anion are very strong bases . Anion are present in two option III AND IV but in anion III N2O group…
Q: Which compound is the strongest base?
A: Strongest base:
Q: Provide the approximate pKa of the following and circle the most acidic hydrogen. a. CH;CH2NH2 b.…
A: The acidic strength is usually represented by pKa. The pKa is the negative logarithm of Ka (acid…
Q: 4. Rank the following compounds in order of increasing basicity. Label the least basic compound "1"…
A: Basicity of a molecule is a measure of the ability to accept proton(H+) to form a stable compound.…
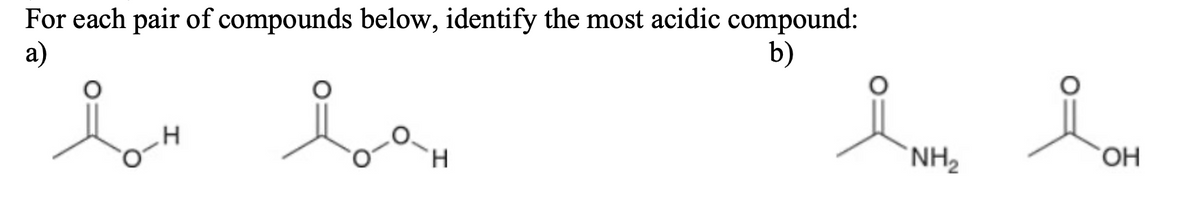
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which of the following is the strongest acid? Group of answer choices CH3OH CH3OH2+ CH3NH2 CH3NH3+NH3 H2O a) Which compound is more acidic and why? b) Which compound has the less stable anion and why?Predict which compound in each of the following pairs of compounds is more acidic and explain yourreasoning for each.(a) HSO4− or HSeO4−(b) NH3 or H2O(c) PH3 or HI(d) NH3 or PH3(e) H2S or HBr
- NH4+ + HAsO4- ⇆ H2AsO4 + NH3What is the Bronsted base in the reaction above? a. NH 4 + b. HAsO 4 - c. H2AsO4 d. more than one response is correctPredict which compound in each of the following pairs of compounds is more acidic and explain your reasoning for each. (a) HSO4- or HSeO4-. (b) NH3 or H2O. (c) PH3 or HI. (d) NH3 or PH3. (e) H2S or HBrWhat is the Base in the reaction below? C2O42– + HC2H3O2 ↔ HC2O4– + C2H3O2– Please choose the right answer: a.) HC2O4– b.) C2O42– c.) HC2H3O2 d.) C2H3O2–
- Rank the given compounds in order of decreasing basicity. 1=most basic, 4=least basicDraw the conjugate base of each of the following acids: a. CH3OH b. CH3NH3+ c. CH3NH2 d. H3O+ e. H2O1.(a) Which of the following groups has the LOWEST IUPAC priority?(A) CH3 (B) NH2 (C) OH (D) COOH (E) Br (b)Which of the following corresponds to the strongest acid?(A) (CF3)3C-COOH (B) (CF3)3 C-OH(C) CH3COOH (D) CH3OH(E) HOCH2CH3
- Caffeic acid is an organic acid isolated from coffee beans. Predict whichlabeled hydrogen (Ha or Hb) is more acidic and explain your choice.Rank the compounds in each of the following groups from strongest acid to weakest acid:1) a) Predict the products of the following acid-base reactions b)draw the electron flow that leads to the products c)indicate wether the equilibrum lies to the left or right of the equlibrium arrow 2) A)for each compound show its conjugate base. Show all valence electrons and any resonance structures b)rank the conjugate base in the order you would predict from most to least stables c)rank the original compounds in order from strongest to weakest acid.

