Chapter17: Alcohols And Phenols
Section17.SE: Something Extra
Problem 54AP: The 1HNMR spectrum shown is that of 3-methyl-3-buten-1-ol. Assign all the observed resonance peaks...
Related questions
Question
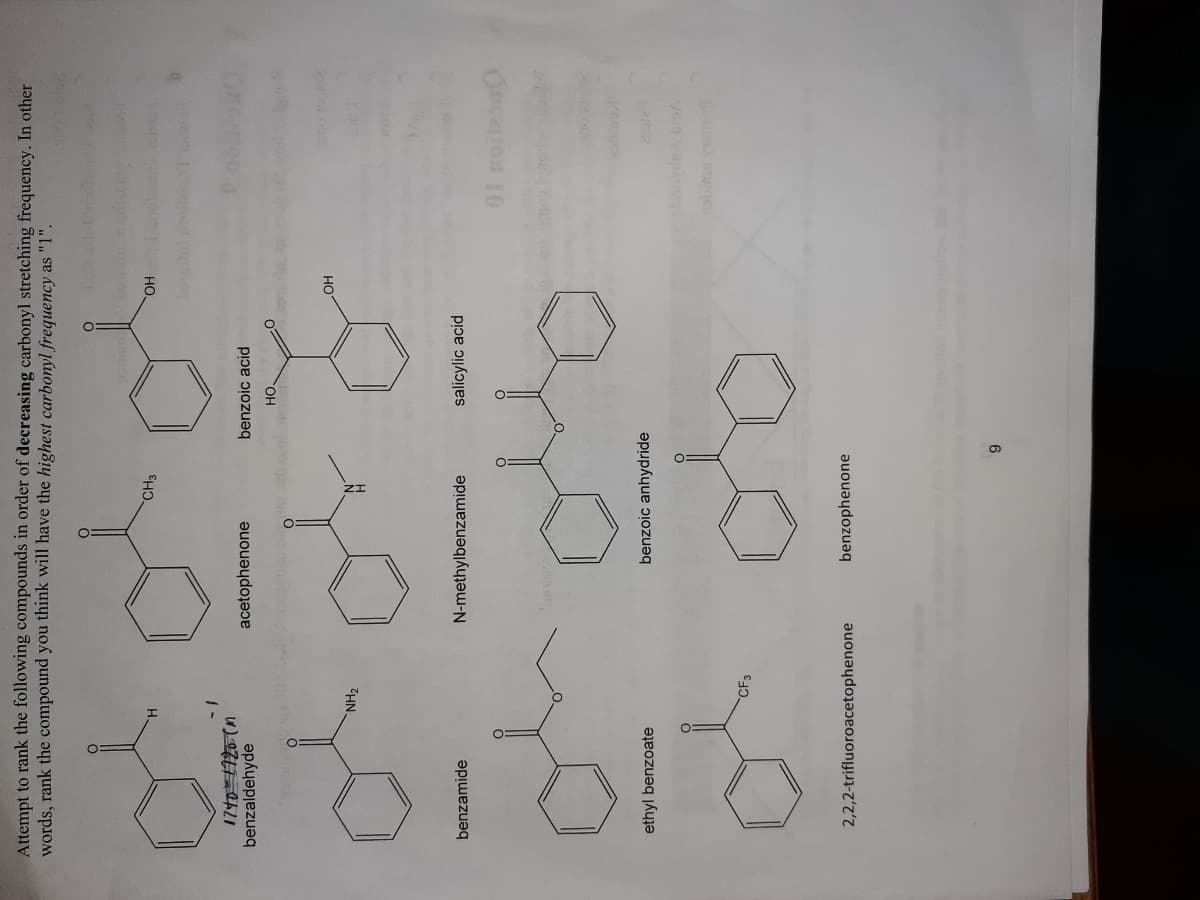
Transcribed Image Text:Attempt to rank the following compounds in order of decreasing carbonyl stretching frequency. In other
words, rank the compound you think will have the highest carbonyl frequency as "1".
CH3
Но
benzaldehyde
acetophenone
benzoic acid
HO.
Но
NH2
benzamide
N-methylbenzamide
salicylic acid
01 moiteoa
ethyl benzoate
benzoic anhydride
CF3
2,2,2-trifluoroacetophenone
benzophenone
6.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

