Q: You are trying to experimentally determine the value for AHreaction. Before that you should…
A: Answer:Hess's law is based on the conservation of energy which suggests that net enthalpy change for…
Q: Complete the following reactions: NaBH₁ LIAIH4 DIBAL, -78°C
A: We are given some reactions, we have to tell their products from different reagent
Q: Based on the results of previous oxidation and reduction questions, complete and balance the…
A: We have to balance the given equation by putting the stoichiometric coefficients in proper…
Q: Consider a 0.292 M aqueous solution of NaClO₂ where MM(NaClO₂) = 90.44 g/mol and the K₂(HCIO₂) = 1.1…
A: We have to choose the correct option.
Q: H* Heat Major Minor
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Consider the following structure: Part 1 of 2 OH How many additional reasonable resonance…
A:
Q: 4. Indicate if each of the following is soluble in water. Explain why or why not. NH₂ CH3-CH-CH3 a.
A: Solubility of amines depends on the ability to form hydrogen bonds with water molecules. It also…
Q: 1. Calculate the fundamental statistical parameters (all measures of central tendency and…
A: Mean (average): Sum of all values divided by the number of values.Median: Middle value when the data…
Q: Which of the following substituted benzenes can undergo Friedel-Crafts alkylation? Select all tha…
A:
Q: Predict the major products of these reactions..
A: The first reaction is the ester hydrolysis reaction in presence of base condition.In the second…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Curved arrows indicate the flow of electrons. The tail of the curved arrow is placed at the source…
Q: Draw the structural formula of benzene -1,3-dicarboxaldehyde.
A: The functional group name is can be identified by the suffix.The position of the functional group…
Q: Which of following statement is incorrect regarding aromatic electrophilic substitution reaction?…
A: A nucleophile is a substance that can donate a pair of electrons. On the other hand, an electrophile…
Q: The simulated APT spectrum of a compound with the molecular formula CH₁, Cl is shown. Draw a…
A: Molecular formula of the compound = C6H11ClThe APT (Attached Proton Test) spectrum for the compound…
Q: What is the value of Ke in boiling water? Click on 'View Image' to use the cheat sheet. a) Ke > 1 c)…
A: When boiling, water is in equilibrium with its vapors. The equilibrium equation for the same is as…
Q: 2. Propose syntheses of the following compounds from benzene: (a) CH3 (b) (c) (d) HO₂S (e) دقة Br…
A: Reactions involves the aromatic electrophilic substitution reactions, diazotization, oxidation,…
Q: What is the name of the following ketone? li r A) 7-methyl-4-ethyl-3-heptanone B)…
A: Select the longest continuous carbon chain.Number the carbon chain from the end in which the carbon…
Q: Draw the mechanisms and draw the major organic product for the following reactions. If indicated,…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 26.12 Poly(3-methyl-1,5-pentanediol adipate) can be prepared by this reaction. Propose a mechanism…
A: Polymers are compounds formed by combining a large number of one or two molecules. The molecules…
Q: Which of the following is miscible in water? Select all that apply: A) NH4 B) CO2 C) CH3CH3 D)…
A: Answer:Polar compounds are soluble in polar solvents and non-polar compounds are soluble in…
Q: Of the compounds shown below, the one numbered enamine H CH3 CH₂CH₂CHCH₂CH₂ acetal || CH₂CH₂CHCH₂CH;…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: he Phe residue drawn as L or D? D L NH2 SH NH ° 2 IN ΗΝ. CH3 OH
A: The objective of the question is to find the configuration of the Phe residue in the given structure…
Q: Predict the major products of the following reaction. Be sure to use wedge and dash bonds to show…
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The…
A: Let me know in comments section if you want to ask any specific question regarding this. I will…
Q: Addition of oxygen nucleophiles: ROH add'n w/acid cat.; prep. of stable acetals 18.13) ● ●…
A: The objective is to determine the mechansim of the Ketone reacting with CH3-OH (methanol).
Q: 4. Predict the major product for the following reaction. HBr a.i H₂C i. Br b. ii c. both i and ii d.…
A: The given reaction is an electrophilic addition to an alkene, following Markovnikov's rule.…
Q: A sample of gas has a volume of 2.50 L at 781 kPa and 75.0 °C. What is the pressure (in atm) of the…
A: The objective of this question is to find the pressure of a gas sample after it expands and cools,…
Q: In the window below draw the structure of cyclohexyl cyclohexanecarboxylate. You do not have to…
A: The carboxylate compounds are compounds that contain the carboxyl group with one photon or hydrogen…
Q: Which of the following Lewis structures represents a better resonance form of [CH3CO]+ than the…
A: Answer:All the atoms in Lewis structure should have complete octet.After the complete octet, if…
Q: The following compound possesses a plane of symmetry. Draw the structure that correctly shows the…
A: I hope this helps you! Thank you! If there's any doubt comment down i will surely be there to help…
Q: An aqueous solution weighing 100.0 g contains 41.9 grams of sodium chloride. What is the mass…
A: The objective of this question is to calculate the mass percent of sodium chloride (NaCl) in an…
Q: 5. Synthesis Design a synthesis of the following transformation. Show how the following multi-step…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What is the OT av U for a gas obeying P = nRT V -a (2) 2
A: The expression represents the rate of change of temperature with respect to volume at constant…
Q: The mass spectrum below belongs to a compound that contains: Relative Intensity 100- 80- 60- 40 20-…
A: Mass spectrometry is an important tool for the determination of the molecular mass of the compound…
Q: 6. Consider the following reaction. CO2(g) + C(graphite) = 2 CO(g) a. A reaction mixture initially…
A: Consider the following reaction: CO2(g) + C(graphite) ⇌ 2CO(g)Kp = 2.25Initially there is 0.56 atm…
Q: Predict the major product of the following reaction: Br Br Br Br HBr (1eq) 0 °C ?
A: In the given question we have to predict the major product of the reaction.The given reactant is an…
Q: The graph represents one mole of a solution. If the heat is added at a constant rate of 225J/minute,…
A: The given graph represents the change in temperature of one mole of a solution over time. It absorbs…
Q: A patient weighing 297 lb is to receive 0.75 mEq/kg of calcium gluconate (Ca(C6H1107)2: MW=430). How…
A:
Q: 3. Provide a mechanism for the following transformation: Ph CI (2 eq.) 2 eq. NaCN 4. Provide a…
A:
Q: An unknown gas effuses at a rate 2.00 times the rate of Kr. What is the molar mass of the unknown…
A: The objective of this question is to find the molar mass of an unknown gas given that it effuses at…
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.20 M…
A: Given data:Different aqueous solutionsDetermine the good buffer.A good buffer consists of:Weak acid…
Q: Write the IUPAC name for the compound below. Be sure to use correct punctuation. CH₂CH3 1 The IUPAC…
A:
Q: Predict the major products of these reaction:
A: The objective of this question is to predict the major products of the given reactions involving…
Q: Using the solubility rules, predict the solubility of each of the following compounds in water.…
A: Answer: 2 (a) CaCO3 : (s) insoluble 3 (b) Al(OH)3 : (s) insoluble 4 (c) Hg2Cl2 :…
Q: Using the Cr value from the below graph find the concentrations of [HCO3], [CO3], [OH], a [H*] at…
A: We have a graph showing the relationship between -log concentration (pH scale) and pH, we can use it…
Q: Draw the Markovnikov product of the hydration of this alkene. Note for advanced students: draw only…
A: The objective of this question is to the Markovnikovs product of hydration of the given alkene.
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of sodium hydroxide…
A: The objective of this question is to write the net ionic equation for the reaction that occurs when…
Q: Which of the following compounds best matches the IR spectrum? LOD
A: IR spectroscopy is mainly used for the identification of the functional group present in the organic…
Q: Which test would best distinguish between the two compounds shown below? Ferric chloride Solubility…
A: Limonene is an unsaturated compound with double bond present.Neomenthol is saturated compound with…
Q: A solution of cobalt chloride of unknown concentration was to be determined using UV-vis…
A: We have to calculate the concentration of unknown solution.
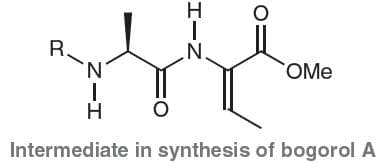
Bogorol A is a natural product with the potential to fight antibiotic-resistant bacteria. Shown below is an intermediate that was used in a synthesis of bogorol A. Assign the configuration of the
a. Z
b. E
Step by step
Solved in 1 steps with 3 images

- To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? a) (1) NaCN; (2) H2O; (3) [H+] b) LiAlH4 c) K2Cr2O7 in acid d) (1) PBr3; (2) NaCN; (3) H2O; (4) [H+]Propose a mechanism for the followingreaction.select the most appropriate reagent(s) to effect the change. 1. Disiamylborane, 2. HO–, H2O, H2O2 H2, Pd K2Cr2O7, H+ NaOCl H2SO4, HgSO4



