R120 General Chemistry F2020 Name: Titration of an Unknown Acid: V, = 25.00 mL Initial Volume Concentration of Final Volume Volume of NaOH (V.) (mL) (V.) (mL) used (mL) V Unknown Acid (M) Flask 1 0.28 25.05 24.77 Flask 2 0.00 25.15 25.15 Flask 3 0.10 25.25 25.15 M Average Acid Concentration: % error: Note: Unknown acid is HCI, so 1 mol HCI gives 1 eq H* for an acid/base titration Given Value: 0.1985 M HCI Calculations: Na. OH caq) + HA caa) → NacI caq) t HzO (e) Average NaOH molayrky: .1410M Conclusion: 1. Where do you think errors could come from in the experiment? 2. How would you modify the experiment to improve % error?
R120 General Chemistry F2020 Name: Titration of an Unknown Acid: V, = 25.00 mL Initial Volume Concentration of Final Volume Volume of NaOH (V.) (mL) (V.) (mL) used (mL) V Unknown Acid (M) Flask 1 0.28 25.05 24.77 Flask 2 0.00 25.15 25.15 Flask 3 0.10 25.25 25.15 M Average Acid Concentration: % error: Note: Unknown acid is HCI, so 1 mol HCI gives 1 eq H* for an acid/base titration Given Value: 0.1985 M HCI Calculations: Na. OH caq) + HA caa) → NacI caq) t HzO (e) Average NaOH molayrky: .1410M Conclusion: 1. Where do you think errors could come from in the experiment? 2. How would you modify the experiment to improve % error?
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 30QAP: Air contains 78% nitrogen. At 25C, Henry's law constant for nitrogen is 6.8104M/atm, while at 37C,...
Related questions
Question
100%
please help lol any help will do! thanks in advance!!
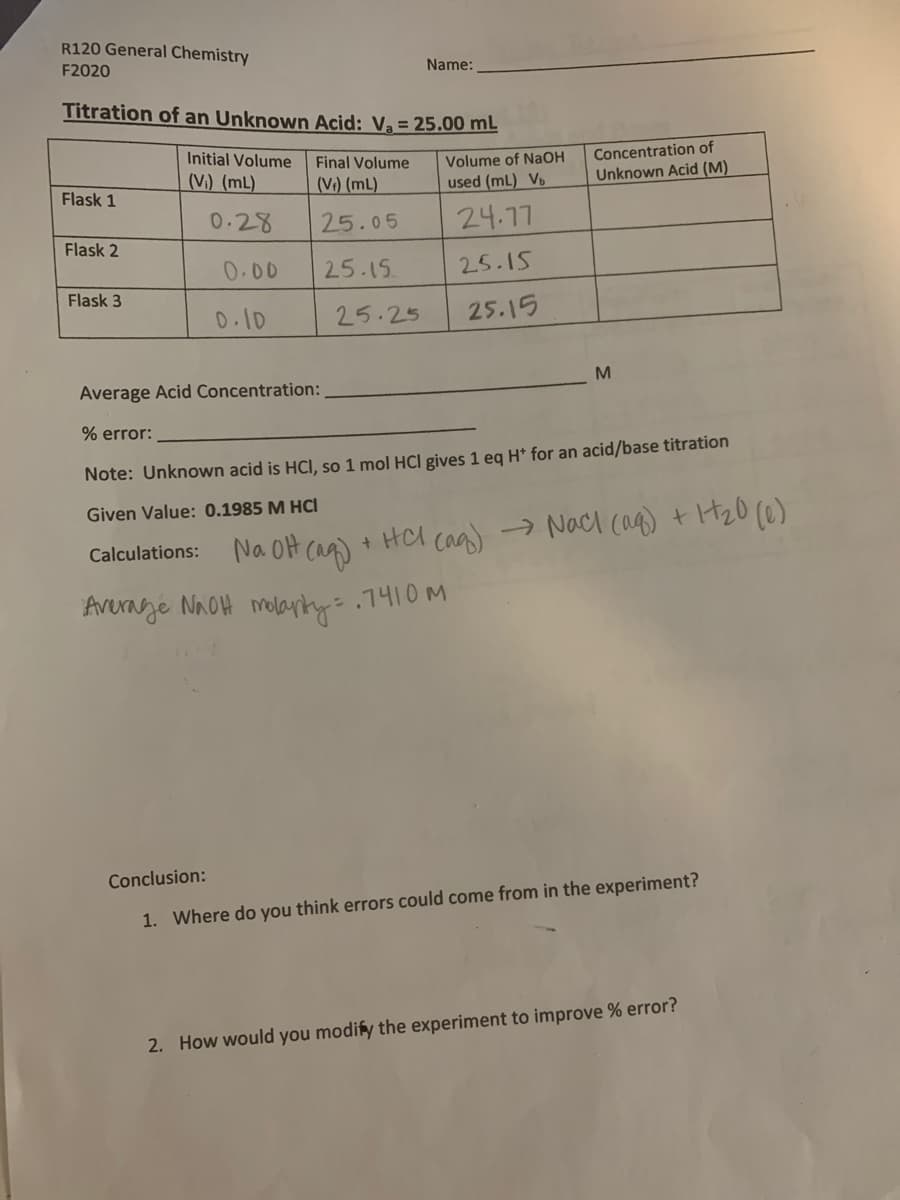
Transcribed Image Text:R120 General Chemistry
Name:
F2020
Titration of an Unknown Acid: Va = 25.00 mL
Concentration of
Unknown Acid (M)
Initial Volume
Final Volume
Volume of NaOH
(V.) (mL)
(V:) (mL)
used (mL) Vb
Flask 1
0.28
25.05
24.17
Flask 2
0.00
25.15
25.15
Flask 3
D.10
25.25
25.15
M
Average Acid Concentration:
% error:
Note: Unknown acid is HCl, so 1 mol HCl gives 1 eg H* for an acid/base titration
Given Value: 0.1985 M HCI
Calculations: Na OH (aq)
+ HA caa) → Nacl cag) t H20 (@)
Avernge NnOH molarty= .7410M
Conclusion:
1. Where do you think errors could come from in the experiment?
2. How would you modify the experiment to improve % error?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning



Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax