Q: A radioactive sample contains 3.01×1018 atoms of a nuclide that decays at a rate of 3.28×1013…
A: The radioactive decay follows first-order reactions. Therefore, N = N0 e -λt
Q: Selenium-73 decays by positron emission with a half- life of 7.15 hours. What mass remains from a…
A:
Q: Barium-142 is an beta emitter with a half-life of 11.0 min. If we start with 2.500 g of 142Ba, what…
A: The number of half-lives (n) can be calculated as
Q: How much time is required for a 6.25-mg sample of 51Cr to decay to 0.75 mg if it has a half-life of…
A: The initial amount of the sample = 6.25 mg The amount of sample after time t = 0.75 mg. The…
Q: The nuclide 3Cl decays by beta emission with a half-life of 37.2 min. A sample of 0.40 mol of H*Cl…
A: Half life of the reaction is the time of the reaction in which the concentration of the reactant…
Q: What is the half-lifeof radioactive element?
A: Breaking down of atomic nuclei of any radioactive substance gives the radiative emissions from…
Q: A radioactive isotope decays from 60 grams to 15 grams in 20 seconds. What is its half-life?
A: Given is the radioactive isotope.We know any radioactive decay follows first order reaction…
Q: -33. The number of radioactive nuclides in a sample decays from 1.00 × 1020 to 2.50 × 10º in 10.0…
A:
Q: 125 Predict the modes of deca of 534 A. Alpha decay B. Positron emission C. Beta decay D. Gamma…
A: Given that : We have to predict he modes of decay of I53125 :
Q: 2) If 20.0 g of a radioactive isotope are present at 1:00 PM and 5.0 g remain at 2:00 PM, what is…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which of the carbon 14 decay by beta emissions
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: billion years after he creetion had he amount of raakletior y, ke om uranium decaypd ne he How…
A:
Q: What is the half‑life of an isotope that decays to 6.25%6.25% of its original activity in 57.1 h?
A: The half-life of the isotopes can be described as the time at which the amount of the activity…
Q: 1. What is the half-life of a radioactive isotope if a 500.0g sample decays to 62.5g in 24.3 hours
A:
Q: Sample of radioactive element with a mass of 200 g, of which 175 g is decayed within 30 days.…
A:
Q: A radioactive element decays at such a rate that after 15 minutes, only 1/10 of the original armount…
A: We will iae intergrated rate equations for first order kinetics to solve this question.
Q: Caesium-137 decays via β - emission with a half-life of 30.17 years. A sample is initially measured…
A:
Q: What is the correct symbol that is missing from the radioactive decay reaction below, and what type…
A: In decay reaction, we have to balance both atomic number and mass number. We know Helium is…
Q: Define Radiotracers?
A: Radiotracers also called as radiolabel compound which are use as a path tracers of reaction , drug…
Q: rbon decays at a rate proportional to the amount present.
A:
Q: Cobalt-60 is a beta-emitter used in radiation cancer treatment equipment in hospitals. What total…
A: Given Initial mass = 24 g half life = 5.33 years time = 16 years
Q: Radium-221 has a half of 30 sec. How long will it take for 82% of a sample to decay?(Round your…
A:
Q: The half life of 52Fe is 8.28 hours. Suppose we measure the activity of a sample containing 52Fe and…
A: Calculation of disintegration constant (λ):T1/2 of 52Fe is = 8.28 hours.
Q: Neptunium-237 undergoes a radioactive decay chain before it reaches nuclear stability, forming the…
A:
Q: Nuclides with too many neutrons to be in the band of stability are most likely to decay by what…
A: Nuclides with too many neutrons lie above in the band of stability. This nuclie which has more…
Q: Cobalt-60 is commonly used as a source of beta-particles. How long does it take for 87.5% of a…
A: Radioactive decay is associated with the emission of small energetic particles e.g. beta particles,…
Q: Cobalt-60, which undergoes beta decay, has a half-life of5.26 yr. (a) How many beta particles are…
A: Half life: It is the amount of time taken by a radioactive material to decay to half of its original…
Q: The half-life of a radioactive element is 6 months. The time taken to reduce its original…
A: Given Half life of radioactive element ( t1/2 ) = 6 month Remaining Concentration ( At ) = 116…
Q: How much of a 0.230 g radioactive sample with a half-life of 8 hours would remain after a period of…
A:
Q: An unknown radioactive element decays into non-radioactive substances. In 340 days the radioactivity…
A:
Q: Isotope X goes through a BETA decay and becomes isotope Y. Isotope Y goes through an ALPHA decay and…
A:
Q: A radioactive sample contains 2.45g. How much willl remain after 11.4 days? (Half life is 3.8 days)
A:
Q: While harder to access another radioactive isotope Tc 93 has a halt life of 2.73 hours of it weee…
A: suppose it is possible to collect Tc 93m, it would be a good substitute because of its high…
Q: 9) Complete the following scheme for 4K decays: (B decay or electron capture or a decay)
A:
Q: c. Luster of 6 Isotope resulted from? a. Natural radioactive decay 6. Substitution of another…
A: Option a) & c) are the correct choices.
Q: Plutonium-239 has a half life of 2.41x10* yr. If you have a 10.0 mg sample how much will remain…
A: The time where any substance/element gets remained just half of its actual quantity is considered as…
Q: 131 For the radioactive isotope 15'I, the time required for 50% disintegration is 8 days. The time…
A:
Q: 14.69 A radioactive sample used in medicine registers an activity of 10 µCi. What is its activity in…
A: Answer: This question is based on unit conversion where we have to convert Radioactivity of a…
Q: 14Cdecays by B-emission and after 12.0 h, ninety percent of the radioactive substance remains.…
A:
Q: Predict the mode of decay of (a) plutonium-239,(b) indium-120.
A: (a) Plutonium-239: The atomic number (Z) of plutonium ( Pu) is 94. So, plutonium-239 has 94 protons…
Q: 27. Which statement is true about the half-life of a radioactive nucleous _____. a. constant for…
A: Half-Life of a radioactive nucleus: The time taken by a radioactive nucleus to decrease its…
Q: A radionuclide undergoes beta decay with half-life of 30.0 years. How many years will it take for 25…
A:
Q: The half-life of sodium-24 is 15.0 hr. How many grams of this nuclide in a 3.00-g sample will remain…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: The half life of radium-226 is 1600 years. How many grams of a 0.25g sample will remain after 4800…
A: After decying the nucleus it emits radiation of electromagnetic rays ,of certain Wavelength.
Q: The radioactive isotope *Pa decays by beta emission. 234 If the mass of a sample of protactinium-234…
A: Initial mass of isotope M = 83.5 ug Final mass of isotope M' = 20.9 ug Time of decay t = 13.5 hours…
Q: A sample of charcoal analyzed in 1995 had an activity of "C = 7.45 dpm g. a. Calculate the age of…
A:
Q: What percentage of Cu-64 will decay in 4.00 h?
A: The half life time of Cu-65 is 12.7 hours. t1/2 = 12.7 hr Radioactive decay is a first order…
Q: Strontium-90 has a half life of 28 years. If a 5.31 mg sample was stored for 112 years, what mass of…
A: Reaction in which rate is linearly depend upon one reactant is known as first order reaction.
Q: The radioactive nuclide Co has a shelf-life of 17.5 hours. What mass Co remains from 0.0100g sample…
A:
40. Which radioactive decay is most probable?
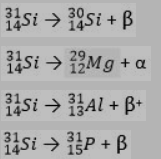
Step by step
Solved in 2 steps

- 3* 7. Correct the errors in the following: 1s22s32p1 1s22s22p63s23d8 1s32s22p63s23p54s1 1s22s22p63s23p63d7What is qrxn? -3661 J -146.4 J +146.4 J +3661 J4 (a) Evaluate four characteristics, and their corresponding considerations of design associated to them for the following reaction: C10H20(liq) + H2(g) = C10H22(liq) ∆Ĥ R < 0 C10H22(liq) + H2(g) = C10H24(liq) ∆Ĥ R < 0
- Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72Calculate delt g for Pbl2(s) + Pb2+(aq) +2I-(aq) T= 25C Kp= 8.7x10-9El poder de rotación específico de la sacarosa a 20 °C es [alfa]D20 = +66,53 grados/(dm g mL-1). ¿Cuál es su poder de rotación molar, expresado en grados/(dm mol L-1)? (Peso molecular de la sacarosa: 342,3). 1). 5.14 2). 22.77 3). 194.4 4). 22773

