Rank the following organic acids in order of acidity, from strongest to weakest. (Hint: consider how resonance and electronegativity will influence the stability of the conjugate base.) Rank from strongest to weakest. To rank items as equivalent, overlap them. Reset Help Н.С — s— он H,C H,C NH, HO,
Rank the following organic acids in order of acidity, from strongest to weakest. (Hint: consider how resonance and electronegativity will influence the stability of the conjugate base.) Rank from strongest to weakest. To rank items as equivalent, overlap them. Reset Help Н.С — s— он H,C H,C NH, HO,
Chapter2: Polar Covalent Bonds; Acids And Bases
Section2.SE: Something Extra
Problem 47AP
Related questions
Question
please help and explain thanks!
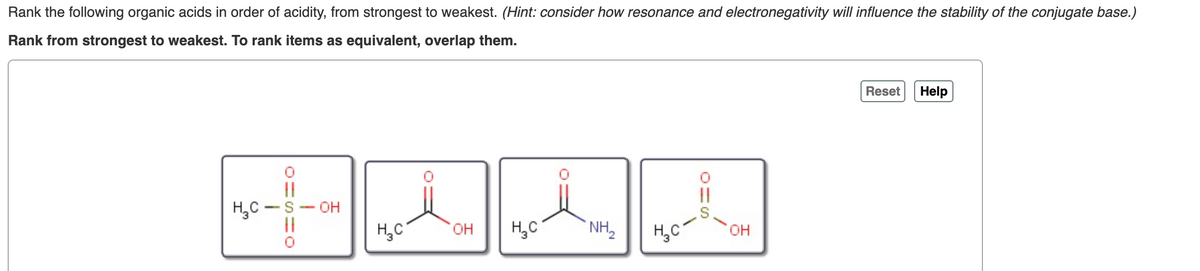
Transcribed Image Text:Rank the following organic acids in order of acidity, from strongest to weakest. (Hint: consider how resonance and electronegativity will influence the stability of the conjugate base.)
Rank from strongest to weakest. To rank items as equivalent, overlap them.
Reset
Help
||
|3|
H,C -S- OH
|3|
H,C
он
H,C
NH,
H,C
он
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning