reaction A B C observations This reaction is always spontaneous, but proceeds faster at temperatures below 121. °C. Crystallization of a pure compound is spontaneous only below 109. °C. This reaction is exothermic and proceeds faster at temperatures above 34. °C. conclusions AH is AS is AH is AS is AH is AS is (pick one) (pick one) (pick one) (pick one) (pick one) (pick one)
reaction A B C observations This reaction is always spontaneous, but proceeds faster at temperatures below 121. °C. Crystallization of a pure compound is spontaneous only below 109. °C. This reaction is exothermic and proceeds faster at temperatures above 34. °C. conclusions AH is AS is AH is AS is AH is AS is (pick one) (pick one) (pick one) (pick one) (pick one) (pick one)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter10: Entropy And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 10.51PAE: 10.51 The combustion of acetylene was used in welder's torches for many years because it produces a...
Related questions
Question
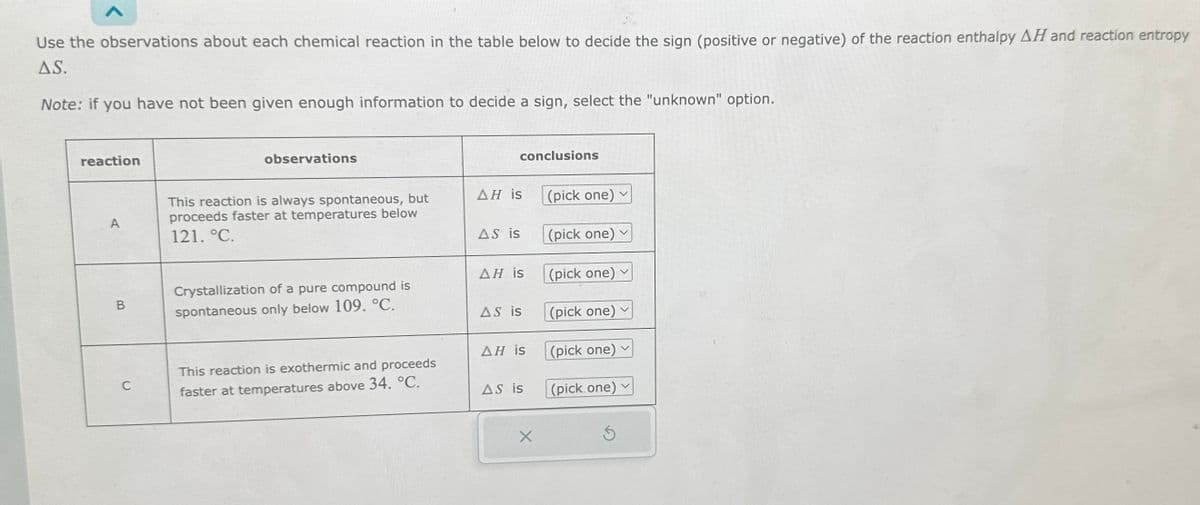
Transcribed Image Text:Use the observations about each chemical reaction in the table below to decide the sign (positive or negative) of the reaction enthalpy AH and reaction entropy
A.S.
Note: if you have not been given enough information to decide a sign, select the "unknown" option.
reaction
B
C
observations
This reaction is always spontaneous, but
proceeds faster at temperatures below
121. °C.
Crystallization of a pure compound is
spontaneous only below 109. °C.
This reaction is exothermic and proceeds
faster at temperatures above 34. °C.
conclusions
AH is
AS is
AH is
AS is
AH is
AS is
X
(pick one) ✓
(pick one)
(pick one)
(pick one)
(pick one)
(pick one)
Ś
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning