Reaction of N2 and Hz in the presence of a catalyst produces ammonia: N2 + 3H2 22NH3 , AH° = -92 kJ mot' . In equilibrium at T= 500°C and p = 200 atm, molar fraction of product is 15% Suggest two ways of increasing molar fraction of product in equilibrium. 1. 12. For reaction of A and B: A + B C, A H° - -20 kJ mol and K = 50 at 25C. Calculate K at 100 C. 13. Using phase diagram for the mixtures of cyclohexane and tolu ene shown, estimate: A) Boiling point of pure cyclohexane B) Boiling point (T) of solution with molar fraction of toluene 80%. c) Composition of vapor phase (molar fractions of both liquids) above the solution with molar fraction of toluene 80% at Tip. D) Compositions of the vapor phase, liquid phase and molar ratio of liquid to vapor phase in the system in which liquid with molar fraction of toluene of 60 % was heat ed to 100 °C at p=1 atm. 14. On the solid-liquid phase diagram for substances A and B shown on left, label all regions of the where solid B coexist with liquid mixture of B and A Mole fraction of B, X Temperature, T
Reaction of N2 and Hz in the presence of a catalyst produces ammonia: N2 + 3H2 22NH3 , AH° = -92 kJ mot' . In equilibrium at T= 500°C and p = 200 atm, molar fraction of product is 15% Suggest two ways of increasing molar fraction of product in equilibrium. 1. 12. For reaction of A and B: A + B C, A H° - -20 kJ mol and K = 50 at 25C. Calculate K at 100 C. 13. Using phase diagram for the mixtures of cyclohexane and tolu ene shown, estimate: A) Boiling point of pure cyclohexane B) Boiling point (T) of solution with molar fraction of toluene 80%. c) Composition of vapor phase (molar fractions of both liquids) above the solution with molar fraction of toluene 80% at Tip. D) Compositions of the vapor phase, liquid phase and molar ratio of liquid to vapor phase in the system in which liquid with molar fraction of toluene of 60 % was heat ed to 100 °C at p=1 atm. 14. On the solid-liquid phase diagram for substances A and B shown on left, label all regions of the where solid B coexist with liquid mixture of B and A Mole fraction of B, X Temperature, T
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter17: Chemcial Thermodynamics
Section: Chapter Questions
Problem 17.95QE: Suppose you have an exothermic reaction with H = 15 kJ and a S of +150 J/K. Calculate G and Keq at...
Related questions
Question
answer #14
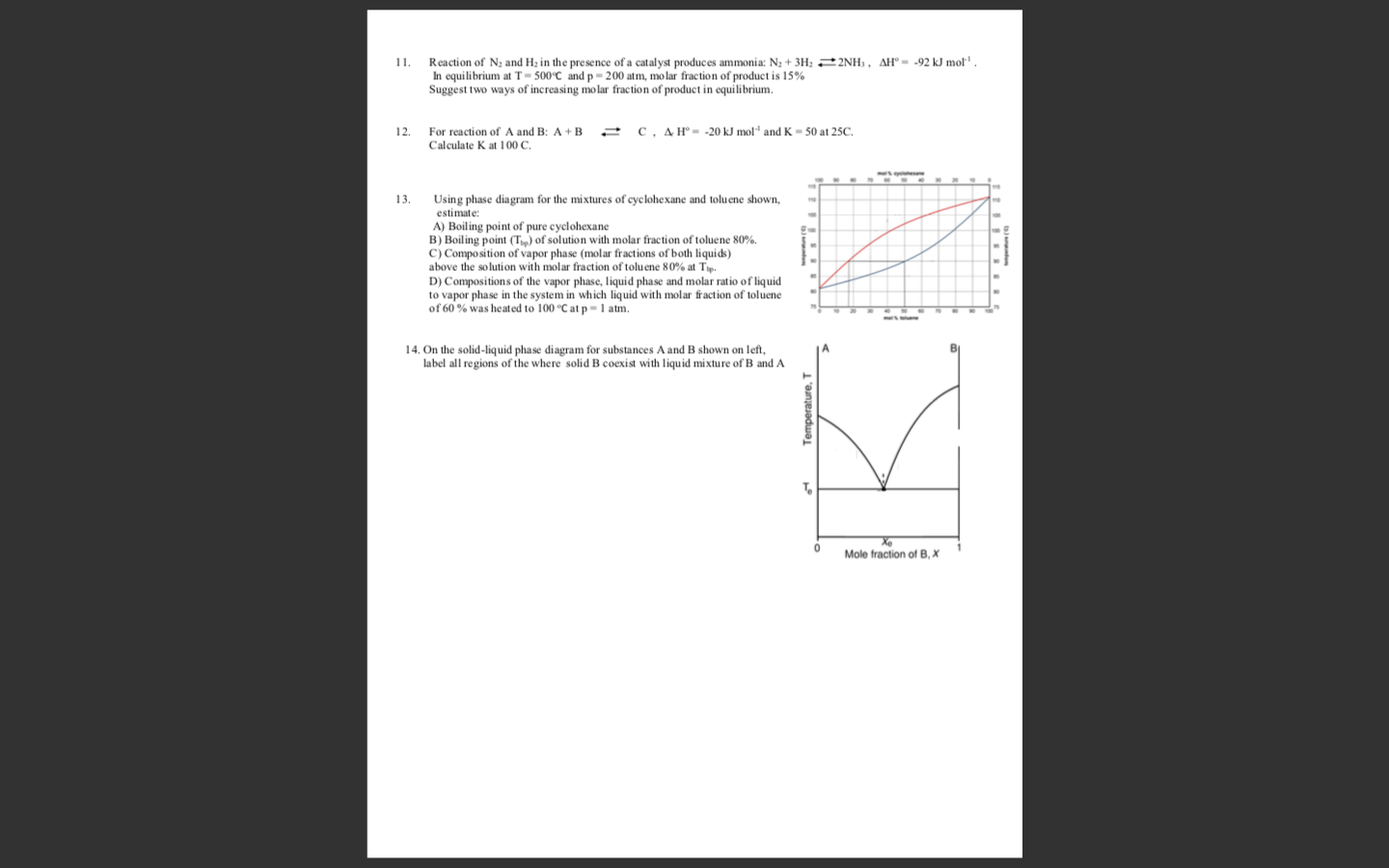
Transcribed Image Text:Reaction of N2 and Hz in the presence of a catalyst produces ammonia: N2 + 3H2 22NH3 , AH° = -92 kJ mot' .
In equilibrium at T= 500°C and p = 200 atm, molar fraction of product is 15%
Suggest two ways of increasing molar fraction of product in equilibrium.
1.
12.
For reaction of A and B: A + B
C, A H° - -20 kJ mol and K = 50 at 25C.
Calculate K at 100 C.
13.
Using phase diagram for the mixtures of cyclohexane and tolu ene shown,
estimate:
A) Boiling point of pure cyclohexane
B) Boiling point (T) of solution with molar fraction of toluene 80%.
c) Composition of vapor phase (molar fractions of both liquids)
above the solution with molar fraction of toluene 80% at Tip.
D) Compositions of the vapor phase, liquid phase and molar ratio of liquid
to vapor phase in the system in which liquid with molar fraction of toluene
of 60 % was heat ed to 100 °C at p=1 atm.
14. On the solid-liquid phase diagram for substances A and B shown on left,
label all regions of the where solid B coexist with liquid mixture of B and A
Mole fraction of B, X
Temperature, T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning