really apologise for posting the same Q please consider this one as I really need it please help me. However, the first image is the one that I did not
really apologise for posting the same Q please consider this one as I really need it please help me. However, the first image is the one that I did not
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 5P
Related questions
Question
I really apologise for posting the same Q please consider this one as I really need it please help me. However, the first image is the one that I did not understand how to do and I really need, and the second one is that what I already have done. so please do the first image like the second that I have done, I mean in the same way but different topic
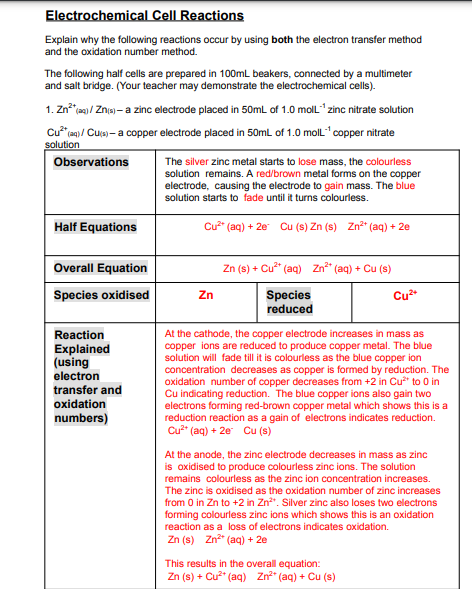
Transcribed Image Text:Electrochemical Cell Reactions
Explain why the following reactions occur by using both the electron transfer method
and the oxidation number method.
The following half cells are prepared in 100mL beakers, connected by a multimeter
and salt bridge. (Your teacher may demonstrate the electrochemical cells).
1. Zn a / Zna - a zinc electrode placed in 50mL of 1.0 moll" zinc nitrate solution
Cu* en)/ Cua – a copper electrode placed in 50mL of 1.0 moll" copper nitrate
solution
Observations
The silver zinc metal starts to lose mass, the colourless
solution remains. A red/brown metal forms on the copper
electrode, causing the electrode to gain mass. The blue
solution starts to fade until it turns colourless.
Half Equations
Cu (aq) + 2e Cu (s) Zn (s) Zn*" (aq) + 2e
Overall Equation
Zn (s) + Cu* (aq) Zn" (aq) + Cu (s)
Species oxidised
Zn
Species
reduced
Cu*
Reaction
Explained
(using
electron
transfer and
oxidation
At the cathode, the copper electrode increases in mass as
copper ions are reduced to produce copper metal. The blue
solution will fade till it is colourless as the blue copper ion
concentration decreases as copper is formed by reduction. The
oxidation number of copper decreases from +2 in Cu* to 0 in
Cu indicating reduction. The blue copper ions also gain two
electrons forming red-brown copper metal which shows this is a
reduction reaction as a gain of electrons indicates reduction.
Cu" (aq) + 2e Cu (s)
numbers)
At the anode, the zinc electrode decreases in mass as zinc
is oxidised to produce colourless zinc ions. The solution
remains colourless as the zinc ion concentration increases.
The zinc is oxidised as the oxidation number of zinc increases
from 0 in Zn to +2 in Zn". Silver zinc also loses two electrons
forming colourless zinc ions which shows this is an oxidation
reaction as a loss of electrons indicates oxidation.
Zn (s) Zn" (aq) + 2e
This results in the overall equation:
Zn (s) + Cu?" (aq) Zn" (aq) + Cu (s)

Transcribed Image Text:Task 2: Electrolysis of Lead Bromide (PbBr,)
White, solid lead (I) bromide is heated until it becomes molten, thus allowing ions to
separate and electricity to flow through it. Once molten, two carbon electrodes,
which are attached to a 12V power supply, are placed in the clear and colourless
molten liquid. Around the electrode connected to the positive terminal, orange/brown
solution is formed At the electrode connected to the negative terminal, a grey solid
formed.
Electrical energy
Electrode:
Electrode:
Graphite/Carbon
Graphite/Carbon
Solution:
PbBr,
2 (aq)
Half equations and overall equation:
Species Oxidised:
Species Reduced:
Explain chemistry involved in electrolytic cell using electron transfer and
oxidation numbers. In your discussion you must:
• Link the observations made at each electrode to the species involved.
• Link half equations for the reaction occurring at the anode and cathode and
identify these as either oxidation or reduction.
• Use standard reduction potentials to calculate the cell potential and predict
spontaneity.
• Link the spontaneity to the relative strengths of the oxidant/ reductant
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

