Reset Help DBAC AG for the reaction Energy of activation for the forward reaction in the Energy of activation for the forward reaction in the presence of enzyme Energy of activation for the reverse reaction in absence of enzyme absence of enzyme
Reset Help DBAC AG for the reaction Energy of activation for the forward reaction in the Energy of activation for the forward reaction in the presence of enzyme Energy of activation for the reverse reaction in absence of enzyme absence of enzyme
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter6: Basic Clinical Chemistry
Section6.5: Blood Glucose And Hemoglobin A1c
Problem 8RQ
Related questions
Question
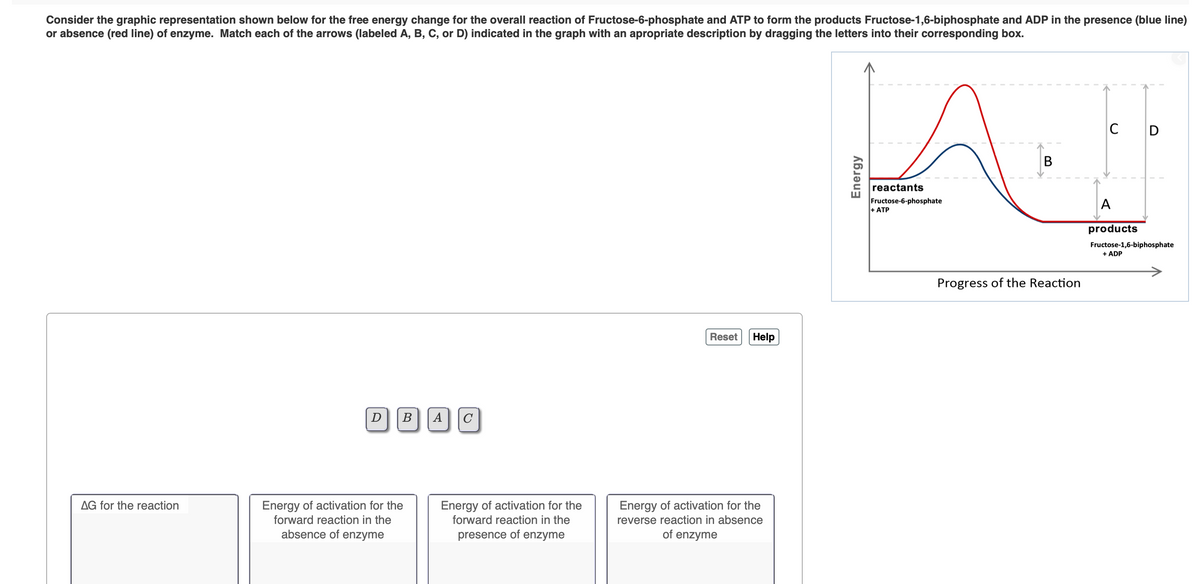
Transcribed Image Text:Consider the graphic representation shown below for the free energy change for the overall reaction of Fructose-6-phosphate and ATP to form the products Fructose-1,6-biphosphate and ADP in the presence (blue line)
or absence (red line) of enzyme. Match each of the arrows (labeled A, B, C, or D) indicated in the graph with an apropriate description by dragging the letters into their corresponding box.
C
D
reactants
Fructose-6-phosphate
+ ATP
A
products
Fructose-1,6-biphosphate
+ ADP
Progress of the Reaction
Reset
Help
D BAc
В
C
Energy of activation for the
forward reaction in the
absence of enzyme
Energy of activation for the
forward reaction
Energy of activation for the
reverse reaction in absence
AG for the reaction
the
presence of enzyme
of enzyme
Energy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

