SECTION 1: ACTIVATION ENERGY The picture to the right shows someone trying to push a boulder up a hill. In order for the boulder to reach its final destination at point B, it must be pushed to the top of the hill because once it reaches the top of the hill it will roll down the rest of the way. A similar scenario exists in chemistry, before a chemical reaction will take place, energy must be put into the system. The energy needed for a chemical reaction to move from point A to the top of the hill is known as activation energy, also written as Ea. Increasing Energy Activation Energy Activation Energy Energy Content of Reactants A + B Reactants FIGURE 3 Progress of Reaction FIGURE 2 Uncatalyzed Reaction (without enzyme) J Catalyzed Reaction (with enzyme) Energy Content of Products C + D Products (TIME) 3. Compare Figure 3 above to the Figure 1 (comparing the heights of 2 mountains), determine how using an enzyme in a chemical reaction impacts the reaction by answering these questions. As seen in the graph a. Does it speed up or slow down the reaction? b. What does the enzyme do to activation energy? c. Based on your answers to questions a and b, what do you think the term "catalyzed" means?
SECTION 1: ACTIVATION ENERGY The picture to the right shows someone trying to push a boulder up a hill. In order for the boulder to reach its final destination at point B, it must be pushed to the top of the hill because once it reaches the top of the hill it will roll down the rest of the way. A similar scenario exists in chemistry, before a chemical reaction will take place, energy must be put into the system. The energy needed for a chemical reaction to move from point A to the top of the hill is known as activation energy, also written as Ea. Increasing Energy Activation Energy Activation Energy Energy Content of Reactants A + B Reactants FIGURE 3 Progress of Reaction FIGURE 2 Uncatalyzed Reaction (without enzyme) J Catalyzed Reaction (with enzyme) Energy Content of Products C + D Products (TIME) 3. Compare Figure 3 above to the Figure 1 (comparing the heights of 2 mountains), determine how using an enzyme in a chemical reaction impacts the reaction by answering these questions. As seen in the graph a. Does it speed up or slow down the reaction? b. What does the enzyme do to activation energy? c. Based on your answers to questions a and b, what do you think the term "catalyzed" means?
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter6: Metabolism
Section: Chapter Questions
Problem 21CTQ: Energy transfers take place constantly in everyday activities. Think of two scenarios: cooking on a...
Related questions
Question
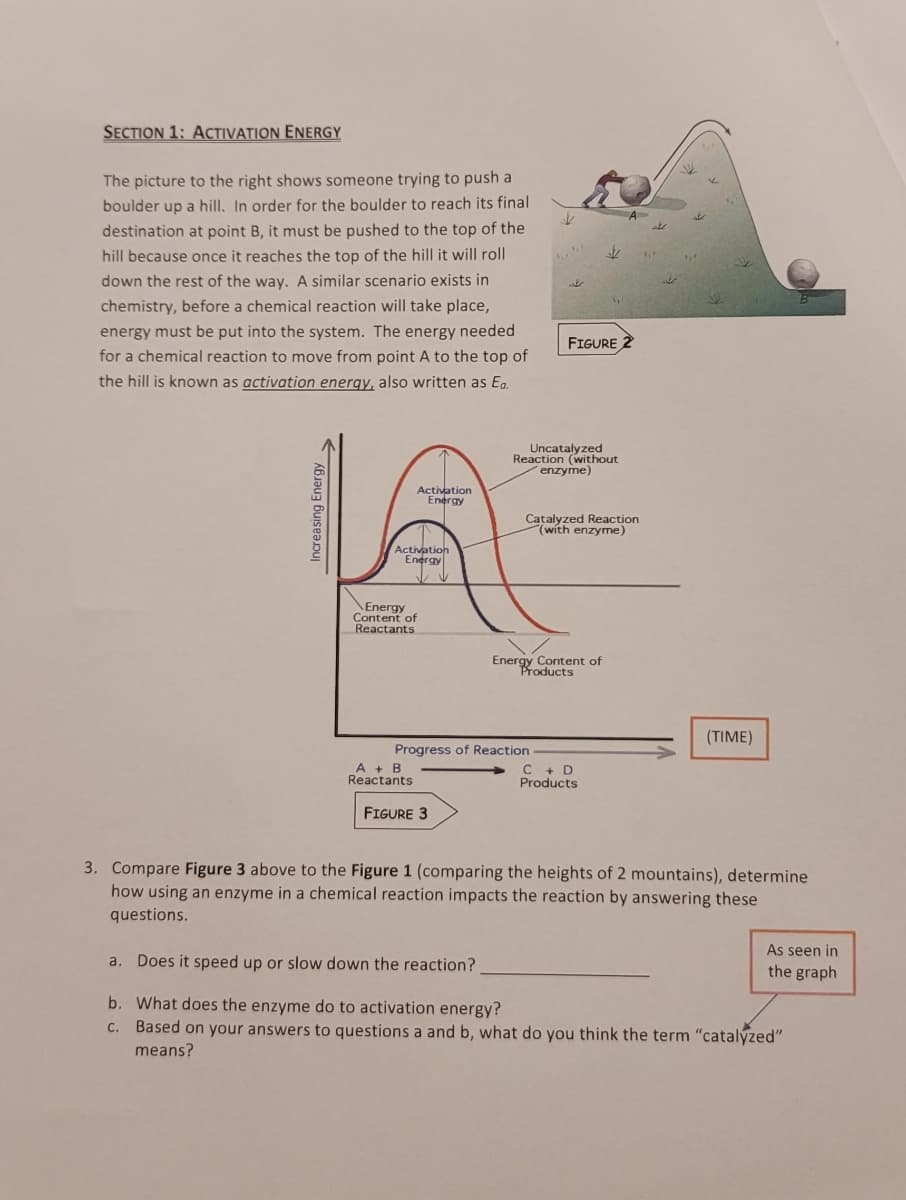
Transcribed Image Text:SECTION 1: ACTIVATION ENERGY
The picture to the right shows someone trying to push a
boulder up a hill. In order for the boulder to reach its final
destination at point B, it must be pushed to the top of the
hill because once it reaches the top of the hill it will roll
down the rest of the way. A similar scenario exists in
chemistry, before a chemical reaction will take place,
energy must be put into the system. The energy needed
for a chemical reaction to move from point A to the top of
the hill is known as activation energy, also written as Ea.
Increasing Energy.
Activation
Energy
Activation
Energy
Energy
Content of
Reactants
A + B
Reactants
FIGURE 3
J
Progress of Reaction
t
$
Uncatalyzed
Reaction (without
enzyme)
Energy Content of
Products
V
FIGURE 2
Catalyzed Reaction
(with enzyme)
C + D
Products
A
1₁²
م
اسکات
(TIME)
3. Compare Figure 3 above to the Figure 1 (comparing the heights of 2 mountains), determine
how using an enzyme in a chemical reaction impacts the reaction by answering these
questions.
As seen in
the graph
a. Does it speed up or slow down the reaction?
b. What does the enzyme do to activation energy?
c. Based on your answers to questions a and b, what do you think the term "catalyzed"
means?
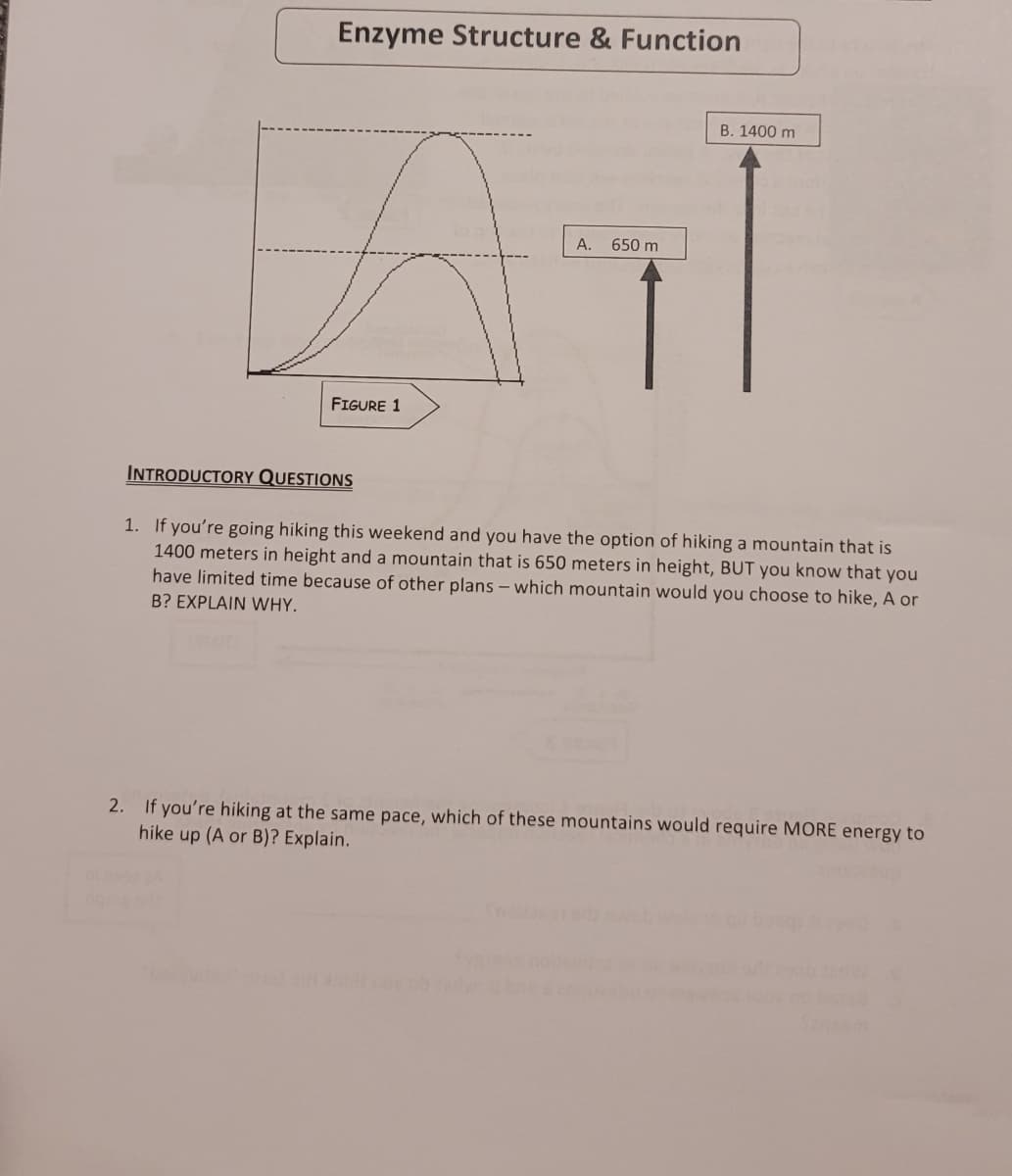
Transcribed Image Text:Enzyme Structure & Function
FIGURE 1
A.
650 m
B. 1400 m
INTRODUCTORY QUESTIONS
1. If you're going hiking this weekend and you have the option of hiking a mountain that is
1400 meters in height and a mountain that is 650 meters in height, BUT you know that you
have limited time because of other plans - which mountain would you choose to hike, A or
B? EXPLAIN WHY.
2. If you're hiking at the same pace, which of these mountains would require MORE energy to
hike up (A or B)? Explain.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax