Chapter9: Alkynes: An Introduction To Organic Synthesis
Section9.SE: Something Extra
Problem 52AP
Related questions
Question
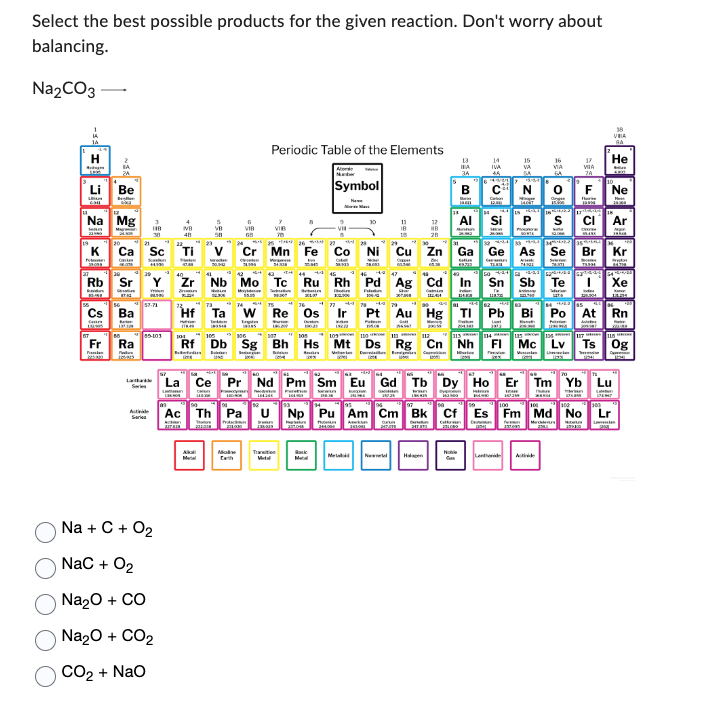
Transcribed Image Text:Select the best possible products for the given reaction. Don't worry about
balancing.
Na₂CO3-
Li Be
LAUR
Oka
Na Mg
230
HOTE
COLLAR
Cs
Caurs
Fr
beiden Seatin wwww.m
re
Ba
Ra
Pake
3
38
Sc
HEM
Series
Rb Sr Y Zr Nb Mo
mais
Na + C + O₂
NaC + O₂
Na₂O + CO
Na₂O + CO₂
CO2 + NaO
4
NVB
48
5
L
58
30.0-0
15-103 104 105
Ta
Akali
BISI
G
VIB
Periodic Table of the Elements
7
VIB
715
Mak
Os
Abode
26 27
Cr Mn Fe Co Ni Cu Zn
TRO
Copper
63.10
2699
54.338
SENES
Symbol
Traiton
Metal
10
4444445
4447
42
Tc Ru Rh Pd Ag Cd
Bebesis
Shar
T
11
IB
Back
28
M
5
Ac Th Pa U Np Pu Am Cm Bk Cf
W Re
INGDO
107
106
105
109 110 111 112
113 114 |
Rf Db Sg Bh Hs Mt Ds Rg Cn Nh FI
defodbo
bidan Snars
13
IIA
34
Ga
2
4-379 44 79
Ir Pt Au Hg Tl
14
IVA
C
12.30
15
VA
N O F Ne
Flare
INGIT
# SURA YAKIN
Al Si P
16
MA
Ge As Se
Sn Sb Te
17
VIIA
7A
La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
okt
HOM
MAR
Lentionide
NE
42-43 244 SAANE
18
VIA
BA
He
Br Kr
indiy
Pb Bi Po At Rn
117 115 tekens
Mc Lv Ts Og
Es Fm Md No Lr
ATDARB
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning