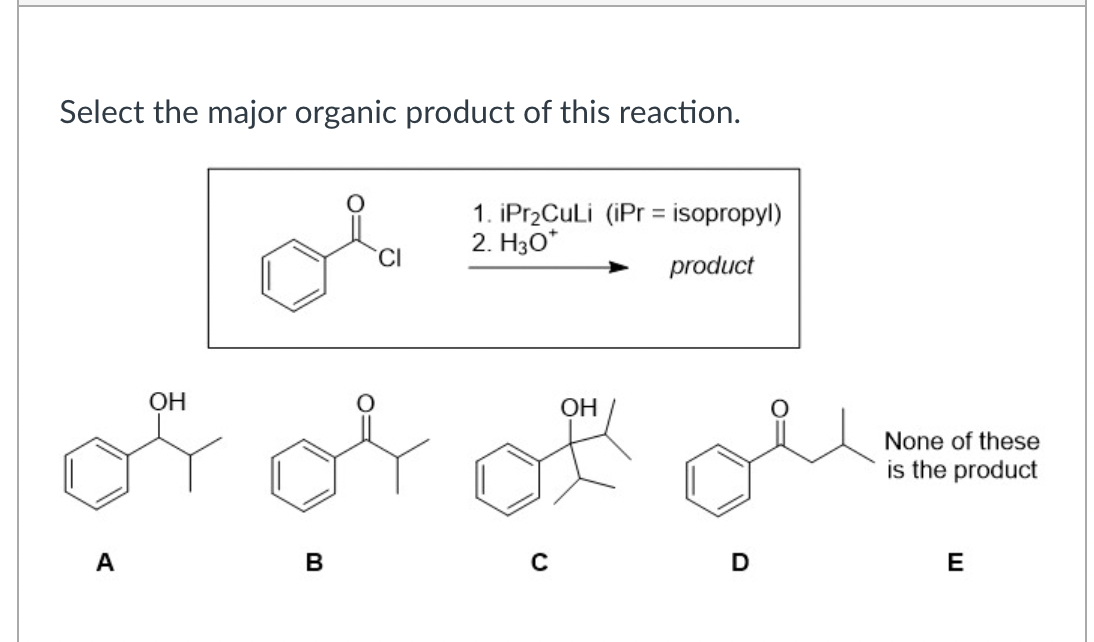
Select the major organic product of this reaction. A OH B CI 1. iPr₂CuLi (iPr = isopropyl) 2. H3O* product C OH D None of these is the product E
Q: Adenosine triphosphate (ATP) is a compound that provides energy for biomechanical reactions in the…
A: Standard enthalpy change () = -20 kJ mol-1Standard entropy change () = 34.0 J K-1 mol-1We need to…
Q: During a recent synthesis of hispidospermidin, a fungal isolate and an inhibitor of phospholipase C,…
A: The mechanism of a reaction shows how atoms and electrons are rearranged between the reactants to…
Q: The enthalpy of solution (AH) of KOH is -57.6 kJ/mol. If 2.91 g is dissolved in enough water to make…
A: Given: Volume of solution = 150.0 mLDensity of solution = 1.02 g/mL.Enthalpy of solution of KOH =…
Q: e b. Ή CH C=C H CH2-CH3
A: Given,The molecule is:b.
Q: why can't you do an ir spectrum or melting point of the product tetraphenylcyclopentadienone being…
A: Benzil (C6H5COC6H5CO) and 1,3-diphenylacetone () are both carbonyl compounds containing two…
Q: Provide the correct systematic name for the compound shown here. H3C CH3 CH3
A: In the naming of the ester, the alkyl or aryl group of the compound will be written first followed…
Q: SnCl₂ has a van't Hoff factor of 2.370. What is the freezing point (in °C) of an aqueous solution…
A: Required data : Freezing point of solution= ?
Q: how many carbon to carbon single bonds, carbon to hydrogen single bonds, number of lone pairs are…
A: The objective of the question is to determine the number of carbon to carbon single bonds, carbon to…
Q: Draw the products of this halogenation rea Use a dash or wedge bond to indicate the stereochemistry…
A: The objective of the question is to draw the curve arrow mechanism for the given reaction in which…
Q: Click on all structures that are enantiomers of the first (leftmost). If no structure qualifies,…
A: In the first step, we assign the substituents priority order based on CIP rules.Next, we see whether…
Q: Arrange the members of the following sets of elements in order of increasing first ionization…
A: Ionization energy :- Energy required to remove an electron from outermost shell of gaseous isolated…
Q: Draw the products of this reaction. Use a dash or wedge bond to indicate the stereochemistry of…
A: alkene compounds are planar and so epoxidation occurs from below and above the planes of alken…
Q: For each of the substituted benzene molecules below, determine the inductive and resonance effects…
A: The objective of the question is to determine the inductive and resonance effects of the CH3…
Q: Write mechanisms for the reactions of 1 - methylcyclopentene with the reagents listed below.…
A: The objective of the question is to understand the reaction mechanisms of 1-methylcyclopentene with…
Q: Why does the pH of the cooking medium influence the texture of cooked fruits and vegetables? What…
A: The pH of the cooking medium can influence the texture of cooked fruits and vegetables due to the…
Q: Give the primary organic product for oxidation of: Select one: O a. No reaction, this compound…
A: What is the primary product of the oxidation of the given alcohol?"
Q: How much did our sodium-in-water demonstration raise the temperature of water in the dessicator jar?…
A: The objective of the question is to calculate the change in energy, in joules, when the reaction of…
Q: Fill in the gaps in the following table. (Enter the symbol with the first (raised) number in the…
A: The number of protons in an atom's nucleus serves as an identifier and characteristic for each one.…
Q: Propose an efficient synthesis for the given transformation. OH This transformation can be performed…
A: A component or combination given to a system to initiate or test a chemical reaction is known as a…
Q: no reaction". For #11 and 1j, work through e final target product.
A: PCC stands for pyridiniumchlorochromate.It is a mild oxidising agent.It oxidizes primary alcohol to…
Q: An aqueous solution contains 0.484 M methylamine (CH3NH₂). How many mL of 0.321 M nitric acid would…
A: The objective of this question is to determine the volume of nitric acid required to prepare a…
Q: Please don't provide handwritten solution ....
A: The Kp for this reaction at the given temperature is approximately 776.13776.13 (unitless)…
Q: What is the p H of a 2.81 M solution of HA that has a Ka of 3.06X10^-5?
A: The objective of this question is to calculate the pH of a solution of a weak acid (HA) given its…
Q: In the determination of the molar mass of a solid acid by titrating it with a standardized base,…
A: The question is what procedural error will cause a solid acid's molar mass to be less than its real…
Q: Draw structural formulas for the a,ß-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: The following product is given:Determine the structural formula of reactants.Gilman reagents are…
Q: What is the IUPAC name of Select one: O a. 1,4-dimethylhexanol O b. 5-methyl-2-heptanol O c.…
A: The parent chain identification is based on the maximum number of carbons in the lengthiest…
Q: Phosphorus pentachloride decomposes at higher temperatures. PCI5(g) PC|3(g) + Cl₂(g) An equilibrium…
A:
Q: ||||...
A: The chair conformation of cyclohexane is considered stable due to its low energy and absence of…
Q: 4. Experimental evidence indicates that the complex [PdCl4]2- has no unpaired electrons, while…
A: The objective of the question is to understand the geometries, oxidation states, and number of d…
Q: CH3 HC-C-OН CH3 1 H₂SO4 H3C H3C C=CH2 2 ?
A: The objective of the question is to understand the reaction between CH3 HC-C-OH CH3…
Q: A 50.0 g sample of unknown metal at 230.0°C is placed in a calorimeter containing 110 g of water at…
A: Heat capacity of calorimeter= 8.2J/g-°cTemperature difference ∆T = 19.3 - 15 = 4.3 °c
Q: Draw the structure of all products of the mechanism below. H H H H H + H Click and drag to start…
A: This is an protonation reaction.. Protonation reaction is the adding of a proton usually denoted by…
Q: (please type answer fast).
A: The objective of the question is to calculate the equilibrium molarity of CO in a reaction where CO…
Q: 4. Given that the molar mass of sulfuric acid is 98.08 g/mol, calculate the molarity of 98% sulfuric…
A:
Q: If the oxidation number of Mn in MnO4 is +7 in the following chemical reaction, then MnO4 is; 5H₂O2…
A: The objective of the question is to determine the role of MnO4 in the given chemical reaction based…
Q: 2. Reductive amination is a great "one pot" method for making substituted amines. Proposed a…
A: In the given question we have to propose a mechanism for the given reaction.The mechanism followed…
Q: Identify the following molecules as chiral or achiral. OH OH Choose... Choose... Choose...
A: For chiral molecule - Their should be the presence of chiral center and no symmetry element should…
Q: Please don't provide handwritten solution ....
A: The objective of the question is to identify the product of a chemical reaction involving CrO3,…
Q: The solubility of Ag2SO4 is measured and found to be 5.16 g/L. Use this information to calculate a…
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [H₂] [¹2]…
A: 1st) Determine the order with respect to H22nd) Determine the order with respect to I23rd) Write…
Q: Consider the following mechanism for the reaction between hydrogen bromide and oxygen: HBr(g) +…
A: An intermediate is a species that is formed in the first step and then reacts in the next step.
Q: Please don't provide handwritten solution .....
A: (a) 10.76 %(b) C10H14OExplanation:
Q: What must be the minimum concentration of cyanoacetic acid in 1.00L buffer solution of a pH 2.97…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: A student has a coffee-cup calorimeter with 50.0 g of water at 21.5oC. They introduce 15.525 g of…
A: The objective of the question is to find the final temperature of the magnesium metal in Kelvin…
Q: What is the absolute configuration (if any) of the following compound?
A: The RS nomenclature is an important nomenclature system for denoting the configuration of the chiral…
Q: Indentify the cooresponding peaks with structure of Aspirin 1H NMR and C13 NMR
A:
Q: Can you please write out the mechanism in detail including all lone pairs, curved arrows, and…
A: Advanced organic chemistry question Explanation:Approach to solving the question: Advanced approach…
Q: A solution of 611.7 ml of 0.00067 M A3B2 is mixed with a solution of 192.5 ml of 0.00099 M C1D2.…
A: Given:- Initial volume of A3B2 solution = 611.7 mL- Initial concentration of A3B2 solution = 0.00067…
Q: What is the primary organic product of hydration of:
A: find out the hydration product
Q: Use the date reported below to graph a cooling plot and use the graphical method to calculate the…
A: Given Data:…
Please don't provide handwritten solution ....
Step by step
Solved in 1 steps with 2 images

- Identify the organic functional group of the product, the reaction type, and predict the functional group of the reactant. Then choose the most likely reactant from the reaction below.The product is a(n)a. aromaticb. alkenec. aldehyded. alcohole. etherThe reaction type is :a. dehydrationb. oxidationc. reduction (hydrogenation)d. hydratione. hydrolysisThe reactant should be a(n)a. aldehydeb. alkenec. esterd. alkynee. alcoholThe most likely reactant is molecule :a. Both C and D would form the productb. D onlyc. Bd. C onlye. AParts A and B are the images, and Part C has the same reactant but H2O and H2SO4 as reagents. Draw the product for each reaction.Name the following alkene and draw the product of the reaction with KMnO4.
- Draw themajor product of this reaction. Ignore inorganic byproducts. PPC, CH2Cl2(CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of theseWhen butane, CH3CH2CH2CH3 and bromine gas, Br2 is exposed to sunlight, monobrominated product are produced. The reaction equation is given below: (i) State the type of reaction (ii) What is the function of the sunlight in the reaction? (iii) Draw the structure of monosubstituted products, A and B. Label the major product. (iv) Draw the propagation steps in the mechanism for the formation of the major product.
- Write the structural formula of the organic product for the given reaction between an alkyne and an alkyl halide. The alkyne group is shown and should be entered as "CC" without the triple bond. Enter CC before associated HH atoms (e.g., CH3CH2CH2OCHCHCH3). what is the name of the product. 1.NaNH2 CH3CH2CCH------> product 2. CH3CH2Brorganic chemistry help Fill in the missing starting materials, reagents, or major products of each reactionFill in any starting materials, reagents, or products to complete the reaction.
- In the reaction from compound 10 to compound 11, why the C=C bond is retained and is not hydrogenated? f) LiAlH4, Et2O, 08C; g) Ac2O, py, DMAP, CH2Cl2, RT, 77% over 2 steps;Complete the following reaction: Pb+2 + SO4 -2 ---------------KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanone

