Q: What is the concentration of citric acid H3C6H5O7 in a 0.400 M solution of citric acid? please…
A: •Concentration:-It is amount of solute dissolved in given amount of solvent or solution. •We use…
Q: Consider the following voltaic cell at 298K. Select only the statements that are TRUE. Cu (s) I Cu²+…
A:
Q: Which statement is true for the condensing of water vapor below 100 °C? O AH is positive; AS is…
A: ∆G shows the spontaneity of a reaction. By considering reaction or process we can figure out sign of…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction free…
A: Given reaction is : 2Al (s) + Fe2O3 (s) ------> Al2O3 (s) + 2Fe (s) Calculate the standard change…
Q: ical formula for formic acid is HCO₂H. To make this buffer, let's say we start with 2.53 M sodium…
A: As per Bartley expert guidelines, I am allowed to answer first one question at a time. Please re…
Q: excess Na₂Cr₂O7/H₂SO4/H₂O
A:
Q: CI J гон A Br NO₂
A: reactions of substituted benzenes
Q: Which substrate concentration range is best for determining the catalytic efficiency of an enzyme…
A: In enzyme catalyzed reaction the catalytic effeciency is defined as how efficiently enzyme converts…
Q: Explain the role and applications of the Cambridge Structural Database CSD (with relevant examples…
A: Cambridge structural database is a repository for metal organic crystal structures and small organic…
Q: When a metal was exposed to photons at a frequency of 1.10 x 1015 s-, electrons were emitted with a…
A:
Q: the question states that the Ka value was 3.0 x 10-8 but in step two to calculate the value of pKa…
A: The Ka of hypochlorous acid HClO in the main question is given by 30.0 × 10-8.…
Q: Chemical Equilibrium: Le Chatelier's Principle Practice Use what you know about Le Chatelier's…
A: The Le chatelier's principle states that , " When any chemical reaction is in equilibrium and a…
Q: Identify the organic class to which the compound belongs based on the functional groups present.…
A: Ans 15. Since R-COOH group belongs to the functional group of carboxylic acid And the above…
Q: Triglycerides are a type of lipid used to store fats. Triglycerides are formed via a condensation…
A:
Q: Given the galvanic cell: SCE || Ag*(aq)|Ag, at 25 °C, and the half-reactions. Agt + e → Ag(s), Eº=…
A:
Q: For some reason, a skier decides to ski from the summit of a mountain near Park City, UT (elevation…
A: Given initial temperature is -17° C and initial pressure is 623 mmHg. Given final temperature is…
Q: Br Mg, 1. CO₂ 2. H3O+ 형 'N H 1. NaOH 2. MeBr
A:
Q: The pH value of a liquid A is 3.8 and the pH value of a liquid B is 6.2. How many times as acidic as…
A:
Q: When you start with 45.6 grams of C6H6, 63.7 grams of C6H5Cl is produced-calculate the percent yield…
A:
Q: What is the geometry of the labeled carbon?? Pyramidal Planar Tetrahedral Trigonal planar *
A: We need to describe the geometry of the pointed carbon.
Q: The actual ionization energy of He is 2372.3 kJ/mol. Which of the following can be said about the…
A: We need to describe the shielding effect in helium.
Q: Identify the unknown compound from the given data:The compound has only 70.5%C, 13.7%H, and 15.6%O.
A: From the given data we have , 70.5% carbon 13.7% hydrogen 15.6% oxygen By the empirical formula…
Q: A chemical compound containing hypothetical elements Av, Bw, Cx, Dy, Ez has mass percentages of…
A: We need to find molecular formula for given compound.
Q: what are some real life examples of thermodynamic studies of Dissolution of Urea? How can this be…
A: Dissolution of Urea : When urea dissolves into the water then bonds between solids breaks and due to…
Q: Determine the Miler indices for the direction represented by an arrow and the planes as shown in…
A:
Q: two representing L-alanine and two representing D-alanine. Which pair represents L-alanine? *H3N-…
A:
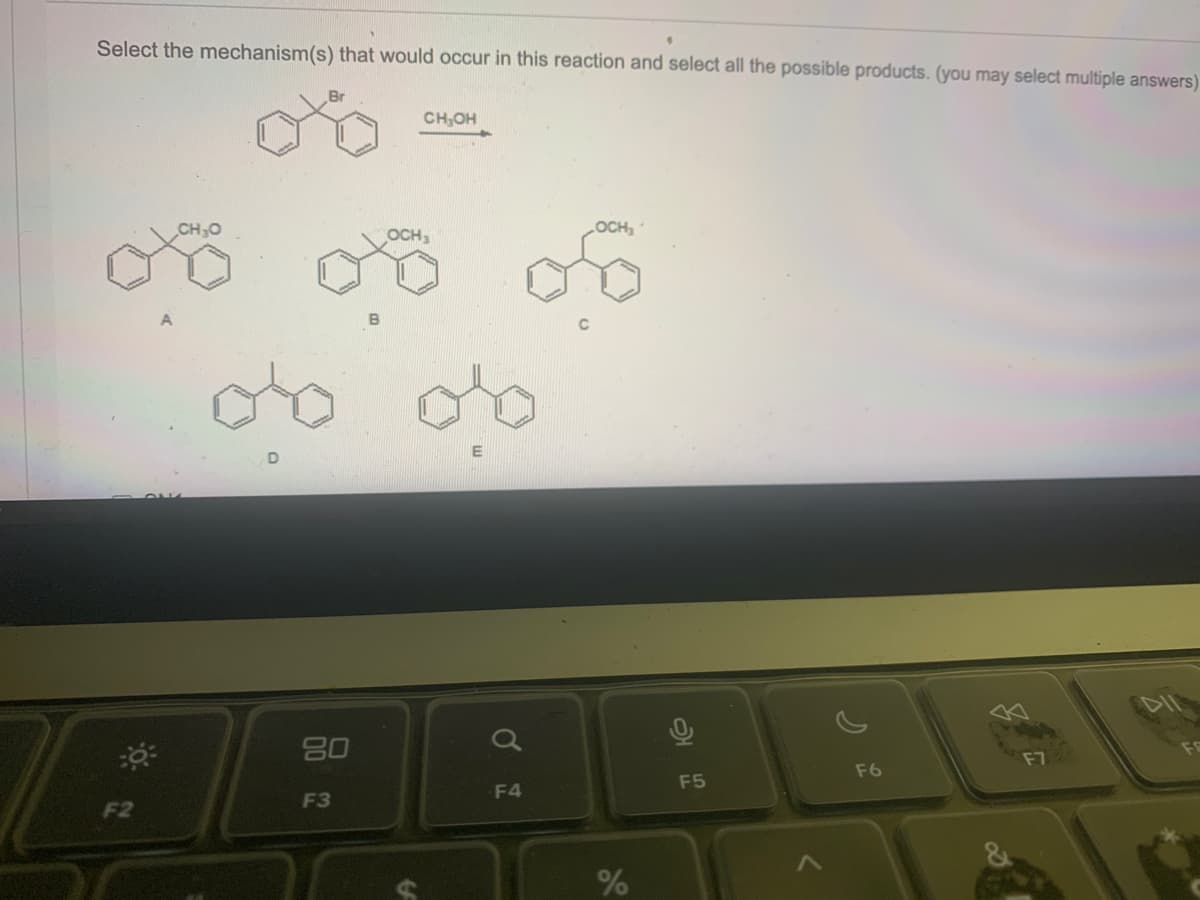
Q: C6H5MgBr + (excess) H3CO OCH 3 H₂O est mỌH OH for H5C6 of C6H5 -C6H5
A: R-MgBr is Grignard reagent. It helps to add alkyl group in a molecule.
Q: Discuss the mechanistic approaches to the degradation of polychlorinated compounds.
A:
Q: A hot lump of 43.8 g of aluminum at an initial temperature of 87.7 °C is placed in 50.0 mL H₂O…
A:
Q: Depict the electron configuration for each of the following atoms using noble gas notation:
A:
Q: For a 35 wt% Sn - 65 wt% Pb composition, draw the schematic of microstructural at the points (solid…
A: The Given 35wt%sb-65wt% pb composition, To draw the schematic of microstructural development at the…
Q: Consider the following reaction: I2(g) + Cl2(g) ⇌ 2ICl(g) Kp=81.9 at 25°C Calculate delta Grxn…
A:
Q: Using noble gas notation, depict reasonable electron configurations for the following element: The…
A: The answer to the following question is-
Q: What is the major product of the following reaction? Br A О с E O A O D О в X Br + Br₂ B heat Br Br…
A: Here we are required to find the major product of the reaction.
Q: Assuming it occurred in an acidic solution, what is the correctly balanced equation for H202 +…
A: (1) H2O2 + Sn2+--- H2O + Sn4+ Balanced in acidic medium ? (2) ∆Go = 12 kJ/mol = 12000 J/mol n = 3…
Q: MISSED THIS? Read Section 5.2 (Pages 168-173); Watch KCV 5.2, IWE 5.2. A laboratory procedure calls…
A: Given, molarity of NaNO3 solution = 1.2 M = 1.2 mol/L Volume of NaNO3 solution = 450.0 mL = 0.450 L…
Q: [Review Topics] [References] Draw both resonance structures of the anion formed by the reaction of…
A:
Q: Which reagents react with an alkene in a Markovnikov orientation? I. HBr II. Br2 III. H20/H2SO4 IV.…
A: To answer Which reagents react with an alkene in Markovnikov orientation.
Q: Identify the missing reagents * A B A H2, Pt Br. u 10 ·} | . Br2, Heat (₂) E Br2, dark O O O Br HBr…
A:
Q: Find the percent by mass of sodium chloride in a 1.20 M NaCl solution. The density of the solution…
A: We knew that % by mass can be calculate by Given formula % by mass = (mass of solute / mass of…
Q: Draw a structural formula for the major organic product of the reaction shown. CH3CH₂ CH₂CH₂CH3 ● H…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.
Q: What are the products for the reaction of iron (III) nitrate and sodium hydroxide? Group of answer…
A: Given reaction reaction is simple inorganic double displacement reaction.
Q: Use appropriate data to determine which statement is true of the voltaic cell picture here.
A:
Q: Fill in the orbital energy diagram for phosphorus. 3s 2s 1s 3p- 2p
A:
Q: Quvon (aq) 2 Fecs) + 3 F₂ cg) → GF - (99) + 2 Fe ² + in which reaction, which is oxidising the agent…
A: Oxidation reduction reaction (Redox reaction) It can be defined as the chemical reaction in which…
Q: Fe(OH)₂ is rather insoluble in water, having Ksp = 1.8*10-15 at 25°C. In order to increase its…
A: •Fe(OH)2 gives (Fe+2) and (OH-) in solution. Fe(OH) => Fe+2 + 2OH- •So if we…
Q: how would you solve this question if it was 60.0ml
A:
Q: Provide the missing reagent to complete the following reaction. CH,CH,CH,ONa ? Write your answer on…
A: Alkyl halide reacts with sodium alkoxide ion to form an ether by the Williamson ether synthesis. The…
Q: a) A solution containing 3.50 g of solute X in 50.0 g of water has a volume of 52.5 mL and a…
A:
Q: What of the following options describes the term "strong acid/base"? O a. A pair of species which…
A: A strong acid or strong base is a strong electrolyte.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The mechanism that allows the exothermic hydrolysis of t-butyl chloride to form t-butyl alcohol and chloride ion is as follows: (CH3)3CCl → (CH3)3C+ + Cl− (CH3)3C+ + H2O → (CH3)3COH + H+ Draw the transition state for each step.Which of the following mechanism types is/are likely in this reaction: CH3 I + KOH → CH3OH +KI i) SN1 ii) SN2 iii) E1 iv) E2What is the molecularity of the rate limiting step for the mechanism below? Step 1: H2O2 (aq) + I–(aq) → H2O(l) + OI–(aq) (slow)Step 2: H2O2 (aq) + OI–(aq) → H2O(l) + O2 (g) + I–(aq) (fast)
- Suppose the formation of tert-butanol proceeds by the following mechanism: step elementary reaction rate constant 1 CH33CBr (aq) → CH33C+ (aq) + Br− (aq) k1 2 CH33C+ (aq) + OH− (aq) → CH33COH (aq) k2 Suppose also k1 ≫ k2 . That is, the first step is much faster than the second.Using the mechanism provided, determine a possible rate law for the following overall reaction: Q2 + 2 AJ2 → 2AJ2Q Step #1. AJ2 + Q2 → AJ2Q + Q slow Step #2. Q + AJ2 → AJ2Q quick Group of answer choices Rate = k [AJ2]2 [Q2] Rate = k [AJ2Q]2 Rate = k [AJ2] [Q] Rate = k [AJ2Q] [Q]2 Rate = k [AJ2] [Q2]Given the following mechanism, which of the species is a intermediate? Choose all that apply. Step1: X+YO2—>XO+YO Step 2: XO+YO2—>XO2+YO Step 3: YO+O2—>YO2+O Step 4: YO+O—->YO2 YO O2 X XO YO2 O XO2
- The following mechanism is proposed: C6H5COOH ⇌ C6H5CO2H2+ (fast) C6H5CO2H2+ ⇌ C6H5CO+ + H2O (fast) C6H5CO+ + HX + H2O ⟶ C6H6NH2 + N2 + CO2 (slow) The overall enthalpy change is -237.6 kJ. Sketch a reaction-energy diagram consistent with the above mechanism and the enthalpy change. Label the enthalpy change and each transition state. Explain the appearance of your graph.2A -> A2 (Fast) A2 + B= A2B (slow) predict the rate law from the following two step mechanismIdentify the overall reaction for the following mechanism. Step 1: NO + NO ⇌ N2O2 Step 2: N2O2 + O2 2NO2
- . The proposed reaction mechanism is as follows:i. BrO3- (aq) + H+ (aq) à HBrO3 (aq) [Fast]ii. HBrO3 (aq) + H+ (aq) à H2BrO3+ (aq) [Medium]iii. H2BrO3+ (aq) + Br- (aq) à Br2O2 (aq) + H2O (ℓ) [Slow]iv. Br2O2 (aq) + 4H+(aq) + 4Br-(aq) à 3Br2 (ℓ) + H2O (ℓ) [Fast]Evaluate the validity of this proposed reaction. Justify your answer.The proposed reaction mechanism is as follows:i. BrO3- (aq) + H+ (aq) = HBrO3 (aq) [Fast]ii. HBrO3 (aq) + H+ (aq) = H2BrO3+ (aq) [Medium]iii. H2BrO3+ (aq) + Br- (aq) = Br2O2 (aq) + H2O (ℓ) [Slow]iv. Br2O2 (aq) + 4H+(aq) + 4Br-(aq) = 3Br2 (ℓ) + H2O (ℓ) [Fast]Evaluate the validity of this proposed reaction. Justify your answer.How are the E1 and E1cB reaction mechanisms different from each other? Draw an example of the reaction mechanism for each one to help illustrate the answer

