Session 21180406 matching question Identify the type of lipid based on the structural features. 1st choice steroids 2nd choice triacylglycerol 3rd choice wax 4th choice sphingophospholipids 5th choice fatty acid contains 3 fatty acids consists of 4 fused carbon rings contains a long-chain alcohol consists of an amide bond consists of only a carboxylic acid and acyl chain Submit response Jump to Send a message to the instructor < Join another session
Session 21180406 matching question Identify the type of lipid based on the structural features. 1st choice steroids 2nd choice triacylglycerol 3rd choice wax 4th choice sphingophospholipids 5th choice fatty acid contains 3 fatty acids consists of 4 fused carbon rings contains a long-chain alcohol consists of an amide bond consists of only a carboxylic acid and acyl chain Submit response Jump to Send a message to the instructor < Join another session
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter2: The Chemical Level Of Organization
Section: Chapter Questions
Problem 26RQ: What organic compound do brain cells primarily rely on for fuel? glucose glycogen galactose glycerol
Related questions
Question
100%
Identify the type of lipid based on structural features.
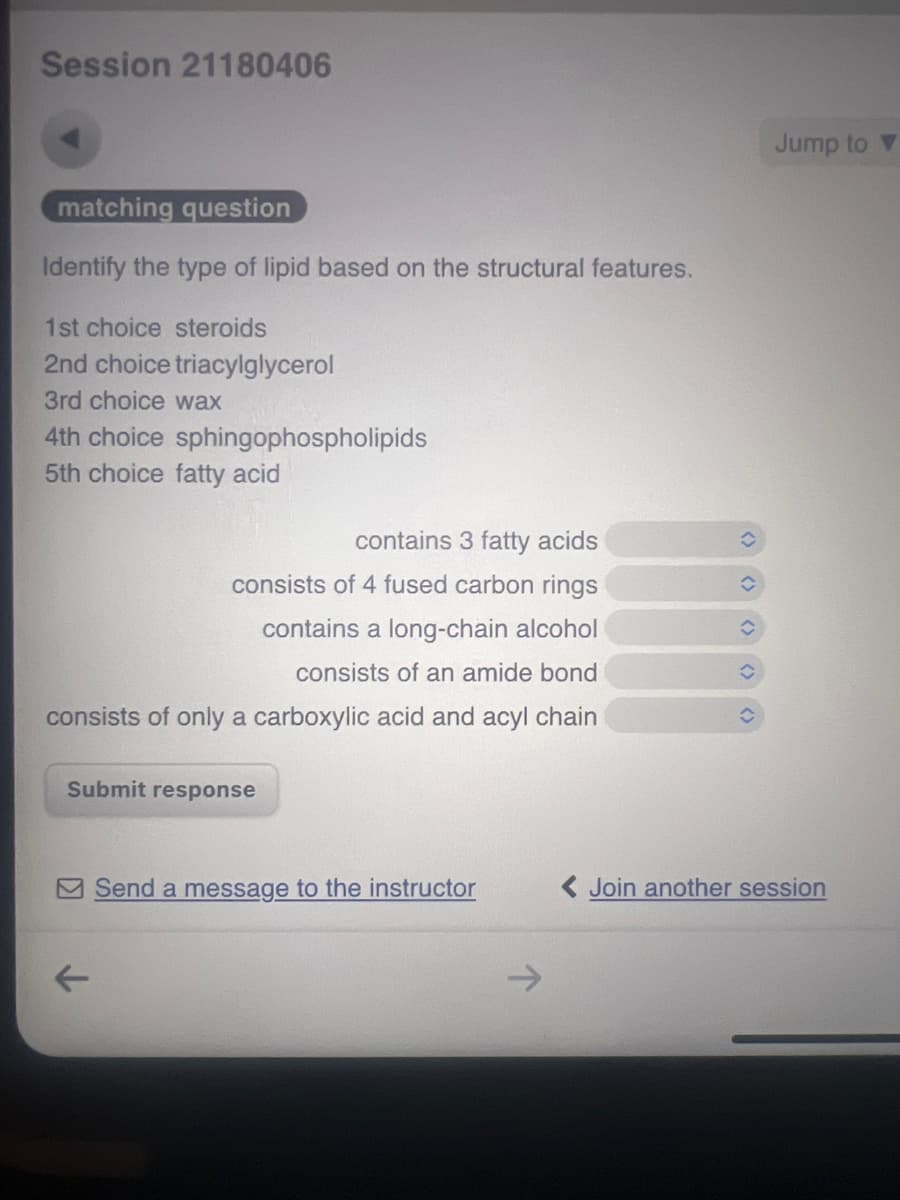
Transcribed Image Text:Session 21180406
matching question
Identify the type of lipid based on the structural features.
1st choice steroids
2nd choice triacylglycerol
3rd choice wax
4th choice sphingophospholipids
5th choice fatty acid
contains 3 fatty acids
consists of 4 fused carbon rings
contains a long-chain alcohol
consists of an amide bond
consists of only a carboxylic acid and acyl chain
Submit response
Jump to▼
Send a message to the instructor
<Join another session
←
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College