Q: (a) * ОН ОН _OCH3
A:
Q: What is the expected major product for the following reaction sequence? CH3 1. BD THF 2. H,O,NAOH OH…
A: Hydroboration–oxidation reaction: Alkene gives an electrophilic addition reaction with borane.…
Q: Complete the following reactions by providing the missing products and reagents A-G. () OH XOH H₂PO4…
A: This is multiple question so I am doing first one as per the guideline if you need all question…
Q: SH S-S 2 + 2HI
A: For the conversion of thiol to disulphides in the lab, we use Iodine in the presence of base and…
Q: For each N-substituted benzene, predict whether the compound reacts faster than, slower than, or at…
A:
Q: 3. The following reaction is used in the industrial synthesis of the antiviral drug, oseltamivir…
A: This problem is based on deprotactation and protection of diols by using different type of…
Q: ベム 1.O3,-78 C 2. DM S
A: The first reaction occurs in the presence of an acid.
Q: When (1R,2R)-2-bromocyclohexanol is treated with a strong base, an epoxide (cyclic ether) is formed.…
A: In the given reaction the alpha bromo alcohol is converted into epoxide in the presence of a base.
Q: Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a…
A: (a) Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence…
Q: но 'N' N.
A: Lithium aluminium hydride will reduce amide into primary amine and after this use PCC to oxidise…
Q: Which set of starting materials could be used to prepare the following compound in one step? * Meo-…
A: Given product is a diels-alder product . In diels -alder reaction conjugated diene reacts with a…
Q: 4. 3. H o-H HotH 5, Br it d,e pair
A:
Q: 2.11. What is the best description of reactions to carry out that will arrive at the following…
A: Synthesis of aldehyde using the right steps.
Q: Rank the following compounds in order of decreasing reactivity towards electrophilic aromatic…
A: For aromatic electrophilic substitution reaction we see electron density where electron density is…
Q: Propose a mechanism (step by step) for the following transformation. Explain each step. HO. H,SO,…
A: Alkenes contain loosely bonded pi-electrons. In the presence of an acid, the alkenes are protonated…
Q: 1. What set of reagents is necessary for the synthesis of the following compound: A A В C D D CN CN…
A: Diels-Alder reaction: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a…
Q: Which of the following alkyl halides would afford the indicated product upon reaction with sodium…
A:
Q: B D OH O,N. OH O,N. NH2 O,N O,N A) Two phenols (A and C) are mixed with oxalyl chloride and…
A: Phenol reacts with oxalyl chloride in presence of triethylamine to form diesters. Here the function…
Q: What products would you expect from each of the following reactions? CH,L/Zn(Cu) diethyl ether…
A: Three questions based on organic chemistry, which are to be accomplished.
Q: Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration. Provide the…
A:
Q: Suggest a mechanism and predict the major product for the following reaction: NABH M OH
A: The reaction is given below: NaBH4 is a mild reducing agent. therefore it only reduced ketone to…
Q: Complete the following reaction sequence from the synthesis of otteliones A and B (Org. Lett. 2003,…
A:
Q: For each compound below, propose an efficient synthesis using diethyl malonate as a starting…
A: (Note- Since it is a multiple questions , hence I am solving first question) Here we have to…
Q: xs. HI
A: This is longer mechanism so read carefully in image First lone pair of oxygen attack on H+ then I-…
Q: (c) Show the mechanistic pathways to give the compound below from 2-methylcyclohex-3- enol. A OH (d)…
A: We have to answer the given questions. C) We have to give the mechanism of the given conversation.…
Q: OMe 11. Me
A: 11. Step 1 : Birch reduction. Step 2 : acid treatment ( double bond isomerization). Step 3 :…
Q: 3) Beginning from acetylene and any alkyl halide needed, how would you prepare the following…
A: Alkynes is easily converted into alcohols by the use of Grignard reagents. Grignard reagents are…
Q: Predict the product and draw a mechanism for the following reaction: 1) LiAlH4 2) H₂O ?
A:
Q: What alcohols would give the following products on oxidation? (a) Он ÓH s od (b) (c) What products…
A: Hi, we are supposed to answer one question. To get the remaining questions solved please mention the…
Q: Chose the reagents necessary to carry out the following conversion: „NH2 `CN LIAIH4 H2O O DIBAL O 1.…
A: 1)LiAlH4 is strong reducing agent it release H2 to readuce any functional group . 2) NaOC2H5 is base…
Q: Select the expected product of the following reaction. Br NaCN DMSO
A: SN2 attack In RDS step two molecules participated Walden inversion product CN- ion approach to…
Q: 4. Propose a reasonable synthesis for the following compound. The starting material is provided. HO…
A: Here we have to convert OH functional group to N3 . So firstly we will make OH leaving group by…
Q: C that can be used as a synthetic detergent to clean away dirt. Outline a mechanism for the…
A: In alkyl benzene, alkyk group is the electron releasing group which increases the electron density…
Q: Propose a synthesis for the following compound starting with benzene. Write out the reagents and…
A:
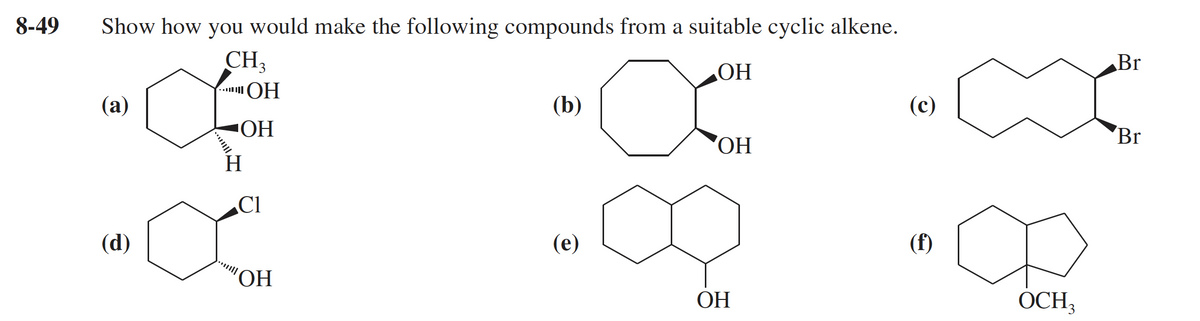
Q: Using any alkene and any reagents necessary, how would you prepare the following compound?
A: Coversion of an alkene to the given alcohol is as follows.
Q: Identify the reagents A in the following scheme: он CHs CHS OH осн CH3 OH NaOCH3 with acid @ low…
A:
Q: (c). Blattellaquinone is the sex pheromone of the female German cockcroach. It is released by…
A:
Q: What is the major product that you would expect to obtain from the following: a.) reaction of…
A: Electrophilic substitution on the substituted benzene ring takes place according to the directive…
Q: NH OH I. I. IV.
A: ->Amine has nitrogen atom with loan pair hence it acts as nucleophile and carbonyl compound has…
Q: NO, H N. `NH2 O,N reagent ОН СНО (A) (B) (С)
A: The correct answer is compound (A). The compound (A) will not react with the reagent beneath.
Q: i) o CH,CH3 • HBr OH Br-CH2CH3 ii) Identify the organic product A, B, C and D in the following…
A:
Q: а) НО Br b) ОН O=
A:
Q: Q3: What products would you expect from the reaction of 1-bromopropane with each of the following?…
A: SOLUTION: Step 1: Hello. Since the question has multiple parts, the first three parts are solved. In…
Q: What products would you expect from the following reaction? OCH3 CI NANH2 liq. NH3
A: Birch reduction involves organic reduction of aromatic rings in liquid ammonia with with sodium and…
Q: How would you synthesize the following compound from benzene? -OH CI
A:
Q: 12. Starting from any alkyne of your choice, suggest a method to synthesis the following products:…
A: Hydration of alkyne: Alkyne gives hydration reactions in presence of an H2SO4 and HgSO4 catalyst. In…
Q: 2) Using any necessary reagents, show how you will accomplish the following synthesis. NH a) b) NH…
A: Given that : We have to synthesize the given products using the necessary reagents :
Q: What would you expect the product of the following reaction to be? BHs: OH OH он NaOOH он A D E F
A: Correct option is D.
Q: The reactivity of the below compounds towards nucleophiles is: R CI R NH2 R OR R b a O a. b>c>d>a O…
A: The compounds are the derivates of carboxylic acid and can be named as: a - Acid chloride b - Amide…
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

- Provide the mechanism and products for the acid-catalyzed epoxide opening reactions below, including appropriate stereochemistry.M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :Complete the flowchart by drawing the resulting structures of each reaction. A. B. C. D. E. F. G. H. I.
- Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below4- Write the complete mechanism of Li/NH3 Reduction of an Alkyne. Show thedirections of the electron flow with the proper arrow orientations. Name the finalProduct.5- What is order of acidity of the following starting from the least acidic to the mostacidic? Alkanes, Alkenes, Alkynes, Aldehydes, Alcohol, Aldehydes, Carboxylic acids.8- automerization is a process in which an enol yields a ketone Give one example for a keto to enol tautomerization. Which form is more stable. Also, which is more stable the E isomer or the Z isomer of 2-Chloro, 2-Butene?
- organic chemistry 7) What is the final product expected from the following reaction?First Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.Dichlorocarbene can be generated by heating sodium trichloroacetate. Propose a mechanism for the reaction, and use curved arrows to indicate the movement of electrons in each step. What relationship does your mechanism bear to the base-induced elimination of HC1 from chloroform?
- The above reaction involves heterolytic bond breakage of HBr a) Encircle the nucleophile (s) and electrophile (s) and explain why. b) Give the mechanism of the reaction by: ) Drawing the appropriate arrows to track the flow of electrons in Showing the species formed after bond breakage and bond formation with appropriate charges. c) Predict the final products.A certain hydrocarbon, C5H6, yields the two products shown below upon ozonolysis with 1. O3/CH2Cl2 2. Zn/H3O . Deduce the structure of the hydrocarbon.Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.

