Some common anesthetics delivered via vaporizer include sevoflurane (200.06 g/mol), desflurane (168.04 g/mol), and isoflurane (184.49 g/mol). Suppose the plot below is for isoflurane. On the same plot, draw the appropriate curves for sevoflurane and desflurane. isoflurane Velocity # of molecules
Some common anesthetics delivered via vaporizer include sevoflurane (200.06 g/mol), desflurane (168.04 g/mol), and isoflurane (184.49 g/mol). Suppose the plot below is for isoflurane. On the same plot, draw the appropriate curves for sevoflurane and desflurane. isoflurane Velocity # of molecules
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter16: Solutions
Section: Chapter Questions
Problem 31E
Related questions
Question
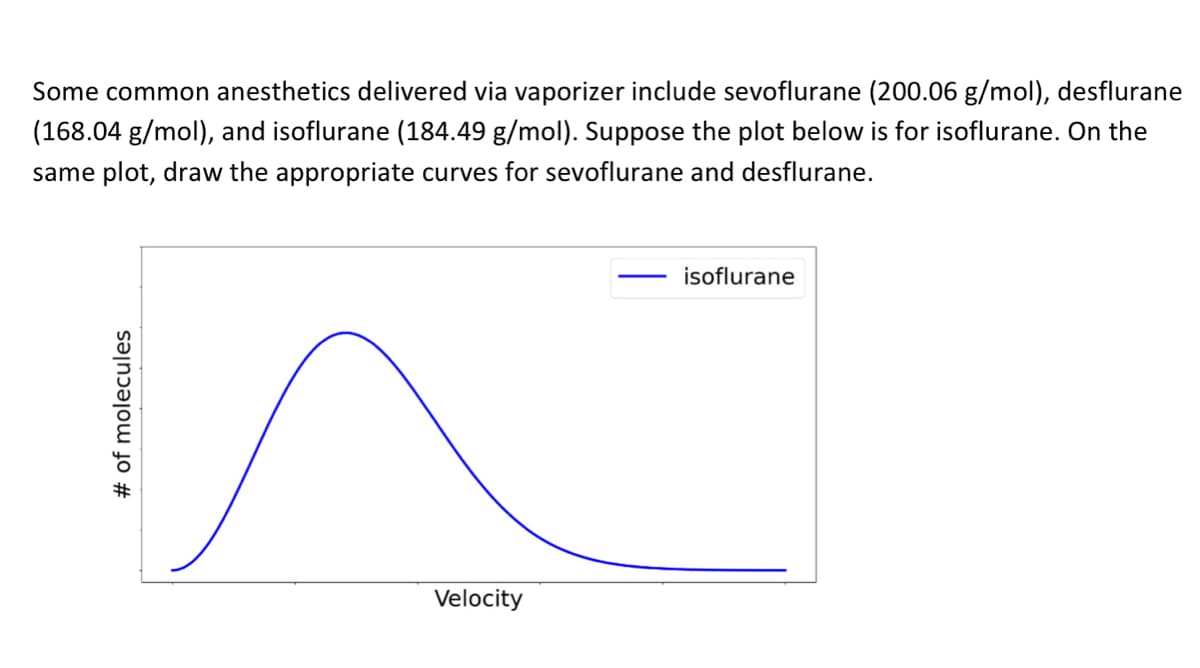
Transcribed Image Text:Some common anesthetics delivered via vaporizer include sevoflurane (200.06 g/mol), desflurane
(168.04 g/mol), and isoflurane (184.49 g/mol). Suppose the plot below is for isoflurane. On the
same plot, draw the appropriate curves for sevoflurane and desflurane.
isoflurane
Velocity
# of molecules
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning