ssohs/3026619/variants/3026619/take/12/ = Governor's Next Generation Commis... >O O< Step 1 2 3 4 5 6 7 Explanation Assign the oxidation number. Split equation into two half- reactions. Identify the oxidized and reduced species. Balance atoms that are not hydrogen or oxygen. Compute electrons lost and gained. Complete the reaction. Check your work. DISMISS Show My Work On the left-hand side, Al is 0, S is +6, and O is -2. On the right-hand side, Al is +3, S is +6, O is -2, and H is +1. Al(s) →Al2(SO4)3(aq) and H₂SO4(aq)→ H2(g) Al was oxidized. H₂SO4 was reduced. You need 2 Al(s) on the left-hand side, and you need 3H₂SO4(aq) also on the left-hand side. 3 electrons lost in oxidation 1 electron gained in reduction You need 3H2(g). This gives 2Al(s) + 3H₂SO4(aq) → Al2(SO4)3(aq) + H2(g)- The aluminum reaction lost 3 electrons and there is one instance of that. The sulfuric acid (H₂SO4) reaction gained 1 electron and there are 3 instances of that. It is balanced. All Changes Saved Continue
ssohs/3026619/variants/3026619/take/12/ = Governor's Next Generation Commis... >O O< Step 1 2 3 4 5 6 7 Explanation Assign the oxidation number. Split equation into two half- reactions. Identify the oxidized and reduced species. Balance atoms that are not hydrogen or oxygen. Compute electrons lost and gained. Complete the reaction. Check your work. DISMISS Show My Work On the left-hand side, Al is 0, S is +6, and O is -2. On the right-hand side, Al is +3, S is +6, O is -2, and H is +1. Al(s) →Al2(SO4)3(aq) and H₂SO4(aq)→ H2(g) Al was oxidized. H₂SO4 was reduced. You need 2 Al(s) on the left-hand side, and you need 3H₂SO4(aq) also on the left-hand side. 3 electrons lost in oxidation 1 electron gained in reduction You need 3H2(g). This gives 2Al(s) + 3H₂SO4(aq) → Al2(SO4)3(aq) + H2(g)- The aluminum reaction lost 3 electrons and there is one instance of that. The sulfuric acid (H₂SO4) reaction gained 1 electron and there are 3 instances of that. It is balanced. All Changes Saved Continue
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 11QAP: When one mol of KOH is neutralized by sulfuric acid, q=56 kJ. (This is called the heat of...
Related questions
Question
Indicate which dots are mistakes ty
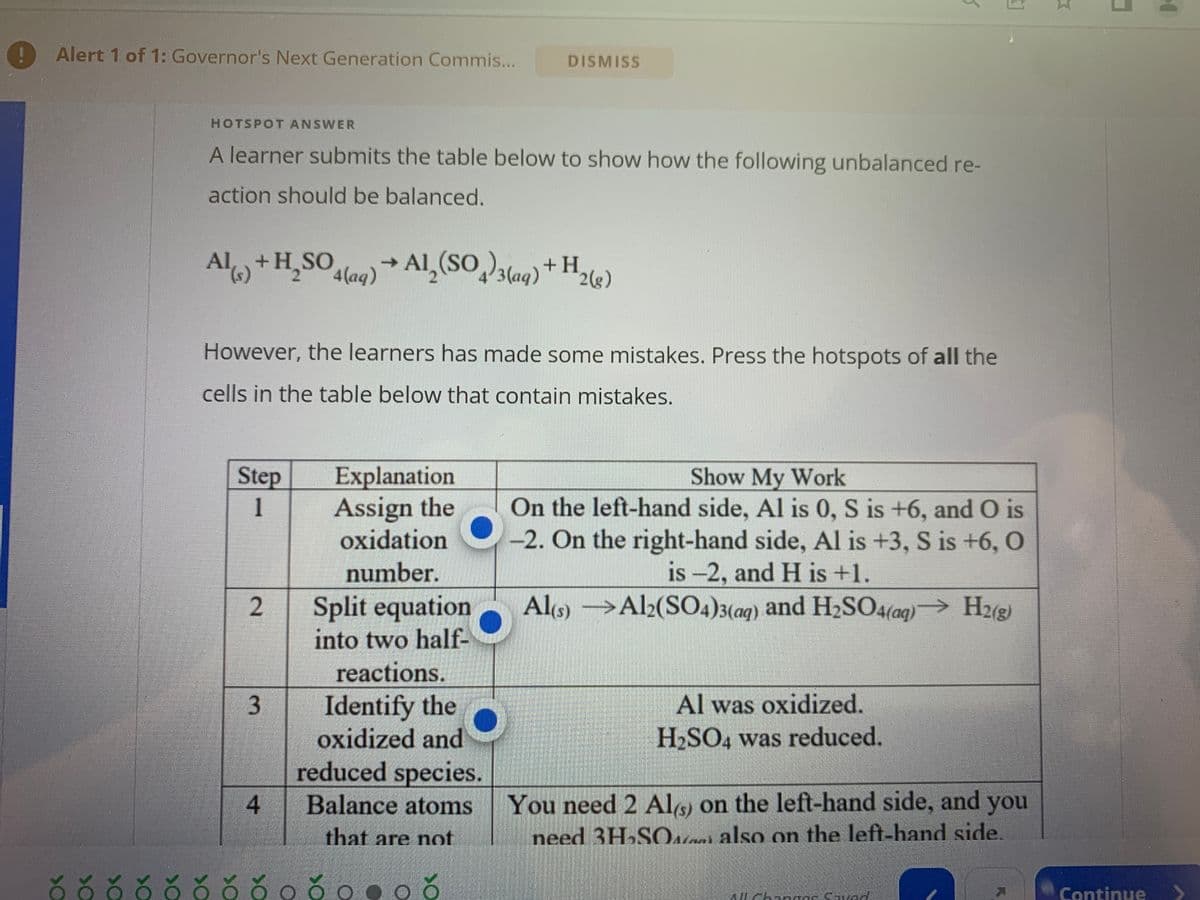
Transcribed Image Text:Alert 1 of 1: Governor's Next Generation Commis...
HOTSPOT ANSWER
A learner submits the table below to show how the following unbalanced re-
action should be balanced.
Al(s) + H₂SO4 (aq) → Al₂(SO4)3(aq) + H 2(g)
However, the learners has made some mistakes. Press the hotspots of all the
cells in the table below that contain mistakes.
Step
1
2
3
Explanation
Assign the
oxidation
number.
4
Split equation
into two half-
reactions.
Identify the
oxidized and
reduced species.
Balance atoms
that are not
á ð ó ó ó ó ó óoðonoð
DISMISS
J
Show My Work
On the left-hand side, Al is 0, S is +6, and O is
-2. On the right-hand side, Al is +3, S is +6, O
is -2, and H is +1.
Al(s)
Al2(SO4)3(aq) and H₂SO4(aq)→ H2(g)
Al was oxidized.
H₂SO4 was reduced.
You need 2 Al(s) on the left-hand side, and you
need 3H₂SO
also on the left-hand side.
K
Continue
De
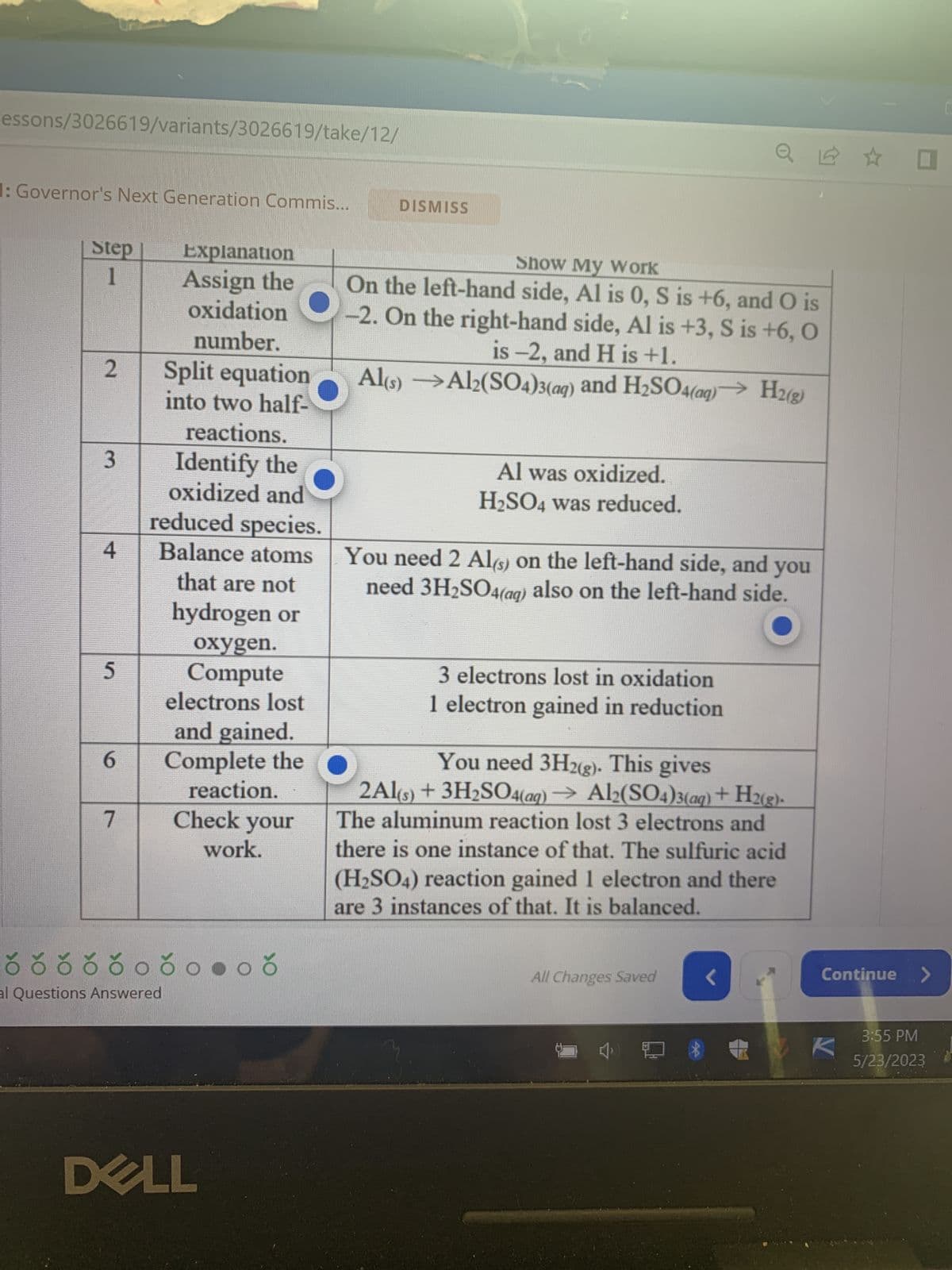
Transcribed Image Text:Alert 1 of 1: Governor's Next Generation Commis...
HOTSPOT ANSWER
A learner submits the table below to show how the following unbalanced re-
action should be balanced.
Al(s) + H₂SO4 (aq) → Al₂(SO4)3(aq) + H 2(g)
However, the learners has made some mistakes. Press the hotspots of all the
cells in the table below that contain mistakes.
Step
1
2
3
Explanation
Assign the
oxidation
number.
4
Split equation
into two half-
reactions.
Identify the
oxidized and
reduced species.
Balance atoms
that are not
á ð ó ó ó ó ó óoðonoð
DISMISS
J
Show My Work
On the left-hand side, Al is 0, S is +6, and O is
-2. On the right-hand side, Al is +3, S is +6, O
is -2, and H is +1.
Al(s)
Al2(SO4)3(aq) and H₂SO4(aq)→ H2(g)
Al was oxidized.
H₂SO4 was reduced.
You need 2 Al(s) on the left-hand side, and you
need 3H₂SO
also on the left-hand side.
K
Continue
De
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning