Q: 3) An unknown solution is subjected to the same flame-test, solubility test, and anion test that you...
A: Since you have asked multiple question, we will solve the first question for you. If youwant any spe...
Q: Arrange the following bonds in order of increasing polarity (least polar first): H---F A...
A: * Greater the electronegative difference greater is polarity. * Electronegativity differene order ...
Q: What is the work done by a system when it absorbs 47 J of heat and gains 12 J of energy? How much he...
A: Since you have posted multiple question , we will solve first one for you. To get the remaining part...
Q: Answer number 5 please
A:
Q: Complete the table, and then analyze the data to determine if Compounds Iand II are the same compoun...
A: The concentration of a substance can be measured in terms of mass by mass percentage. This concentra...
Q: Answer number 4 please put in graph format
A: Given data,Volume of CO2=0.5LThe reaction can be given as;4C3H5 + 17O2→ 12CO2 + 10H2O
Q: Determine whether each molecular diagram represents a pure substance or a mixture. If it represents ...
A: Element is the pure form of matter which only consists one type of atom. A compound consists molecul...
Q: Determine the number of significant figures in each measurement. a. 508.0 L c. 1.0200 × 1...
A: Significant figures refer to the number of digits that are involved in the determination of the degr...
Q: What is the mass of a 1.75 L sample of a liquid that has a density of 0.921 g/mL? a) 1.61 x 10'g c) ...
A: Density is given by the ratio of mass of the substance to that of the volume.
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Amino acids: Amino acids are monomers of a protein. Amino acids through peptide bonds protein molecu...
Q: Show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: All of the given compounds have same molecular formula i.e C5H11OH but different structures, so thes...
Q: 2. Provide the reagent(s) that are needed to perform the oxidation transformations given below. a. C...
A: a) PCC(pyridinium chlorochromate) b)potassium dichromate in the presence of dilute sulfuric acid
Q: Show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Dear student I have given answer to your question in the image format .
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer
A: Transition elements are those having partially filled d orbitals.
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Strong acids and bases are the ones which undergo complete dissociation.Weak acids and bases are the...
Q: An object with a mass of 7.5 g raises the level of water in a graduated cylinder from 25.1 mL to 30....
A: Density of the object is given by the ratio of mass of the substance to that of the volume of the su...
Q: Chemistry question
A: Reaction 1 we have given the alkyne substrate which on addition of excess HBr give geminal dihalide.
Q: What three elements are found in chlorofluorocarbons?
A: The three elements that are found in chlorofluorocarbons has to be given.
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Atoms of isotopes of the same element are identical in atomic no but different mass no . Atomic no e...
Q: Predict the major product of the reaction below. HNO, H,SO4
A: Interpretation - To predict the major product in the given reaction which is given in the question ...
Q: Show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A:
Q: The structure of vinyl chloride is shown. vinyl chloride This alkene undergoes addition polymerizati...
A: Polymerization is a process by which there is a large number of units of an alkene and alkyne which ...
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: When butan-2-ol is heated with concentrated H2SO4 , but-2-ene is the major product according to Sayt...
Q: Show the product(s) of the following reaction. 1. NAOH 2. NH,CI
A: Ester hydrolysis reaction: In the ester hydrolysis reaction, the ester is converted into carboxylic ...
Q: How is acid strength related to the direction of equilibrium in an acid–base reaction?
A: To find: How is acid strength related to the direction of equilibrium in an acid–base reaction
Q: Use Figure 2.19 to answer the following questions.a. Which substance has the greatest density?b. Whi...
A: Given figure : In above figure , density of material showing on vertical axis. And material name sh...
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Transition elements :- The elements which consists of partially fill d-orbitals either in ground sta...
Q: Quantum numbers arise naturally from the mathematics used to describe the possible states of an elec...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: A sample of gas contains 4.19 moles at 37.8 °C in a volume of 3.61 L. At what pressure is this gas, ...
A:
Q: 1) Balance the following in acidic solution: a) VO2* + Zn → VO2* + Zn2+ b) Fe2*(aq) + MnO4 (aq) → Fe...
A: BALANCING EQUATION IN ACID SOLUTION A) VO2+Zn→VO+2+Zn+2 STEP 1 IDENTIFY THE OXIDATION STATES V in ...
Q: What is the molarity of an HCl solution if 22.5 mL of a 0.100 M NaOH solution are needed to titrate ...
A: The balanced chemical equation for the reaction of HCl and NaOH is as follows: HCl + NaOH ↔ H2O + Na...
Q: Show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: A multiple choice question based on intermolecular force, which is to be accomplished.
Q: Convert each temperature. a. 212 °F to °C (temperature of boiling water at sea level) b. 22 °C to K ...
A: a. 212 °F to °C (temperature of boiling water at sea level): T(°C)5=T(F)-329 T(°C)5=212-329 =1809=20...
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Uncatalysed reaction activation energy is 160 kJ/mole Decrease activation energy after using ...
Q: Answer number 1 please
A: Chemical Reaction:- 4 Fe + 3 O2 --- > 2Fe2O3 4 mol of iron atom react with 3 mol o...
Q: 4 Fe +3 O2 ---> 2 Fe2O3 How many litters of iron metal are needed to react completely with 4.35 m...
A: A numerical problem based on mole concept, which is to be accomplished.
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: To find out this we need to consider electronic configuration of magnesium. Electronic configuration...
Q: 8. Explain WHY the reaction of alkene with water/alcohol require acid, while the reaction of an alke...
A: Answer Reaction of water/ alcohol require acid but Br2 not becau...
Q: What is a derived unit? List an example.
A: There are seven basic SI units given by the international system of units. All the other units come ...
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: 46. The first statement implies to A that is the given statement is the correct explanation of the g...
Q: *Please explain the steps and write the cathode and anode half-reactions, thank you in advance* Co...
A: Interpretation - To explain the steps and write the cathode and anode half cell reaction for the el...
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer keyboa...
A: Since we know that one mole of any substance contains 6.022 × 1023 molecule And Number of mole = m...
Q: A 5-mL sample of water has a mass of 5 g. What is the density of water?
A: The given data contains, mass = 5 g. Volume = 5 mL.
Q: Give answer last three questions with explanation please
A: The reactions given by the H2SO4 and DCM are named as acid-base reactions. Acid tend to release its ...
Q: Why chemists use models to study atoms?
A: An atom is composed of three component particles which are electrons, protons and neutrons. The pro...
Q: Classify each as a physical change or a chemical change.a. breaking a pencil in twob. water freezing...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Cell Notation Diagram, identify the two half-reactions occurring and the overall redox reaction Fe(s...
A: Fe(s)|Fe2+(aq)||Ag+(aq)|Ag(s) At anode:- Fe ----------> Fe+2 +2e- At Cathode:- 2Ag+ +2e- -----...
Q: Balance the following chemical reaction.
A: A balanced equation is the equation in which equal number of atoms are present on both the sides of ...
Q: 3. Explain the process of sublimation Name one substance that sublimes at room temperature and press...
A: A substance can exist in various forms such as solid, liquid, and gas. The conversion of a solid int...
Q: Gamma-ray photons have: O no mass and an electric charge of +1 no mass and an electric charge of -1 ...
A: Gamma rays (γ) are photons.
Show-all-working-explaining-detailly-each-step.
Answer should be typewritten using a computer keyboard
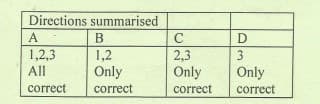

Step by step
Solved in 2 steps with 1 images

- Giventhefollowingsetofreactionsandtheirenthalpies,drawtheBorn-Habercyclefortheformationof LiCl(s)anddeterminethelatticeenergyforLiCl.Reactions:H (in kJ)(1)Li(s)Li(g);Hs= 161;(2) Li(g)Li+(g)+e–;IE= 520;(3) ½Cl2(g)Cl(g);½BE =121.5;(4)Cl(g)+ e–Cl–(g);EA= –349;(5)Li+(g)+ Cl–(g)LiCl(s);UL= ?(UL= latticeenergy)(6) Li(s)+ ½ Cl2(g)LiCl(s);Hof=–408.6._______________________________________________________________________________5.) Which of the analine "units" in the Structure below would have the lowest pka which would have the highest A B H₂N. H₂N. O IZ NH₂ — с NH, D andThe electronic configuration of 15 X 3- ion is a. [Ne]3s23p2 b. [Ne]3s2 c. [Ne]3s23p6 d. [Ne]3s23p1

