Strontium chloride and sodium fluoride react to form strontium fluoride and sodium chloride, according to the reaction shown. SrCl, (aq) + 2 NaF(aq) → SrF, (s) + 2 NaCI(aq) What volume of a 0.390 M NaF solution is required to react completely with 795 mL of a 0.460 M SrCl, solution? volume: mL How many moles of SrF, are formed from this reaction? moles of SrF: mol
Strontium chloride and sodium fluoride react to form strontium fluoride and sodium chloride, according to the reaction shown. SrCl, (aq) + 2 NaF(aq) → SrF, (s) + 2 NaCI(aq) What volume of a 0.390 M NaF solution is required to react completely with 795 mL of a 0.460 M SrCl, solution? volume: mL How many moles of SrF, are formed from this reaction? moles of SrF: mol
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter3: Chemical Reactions
Section3.11: Stoichiometry In Aqueous Solutions
Problem 3.28E: Sodium chloride is used in intravenous solutions for medical applications. The NaCl concentration in...
Related questions
Question
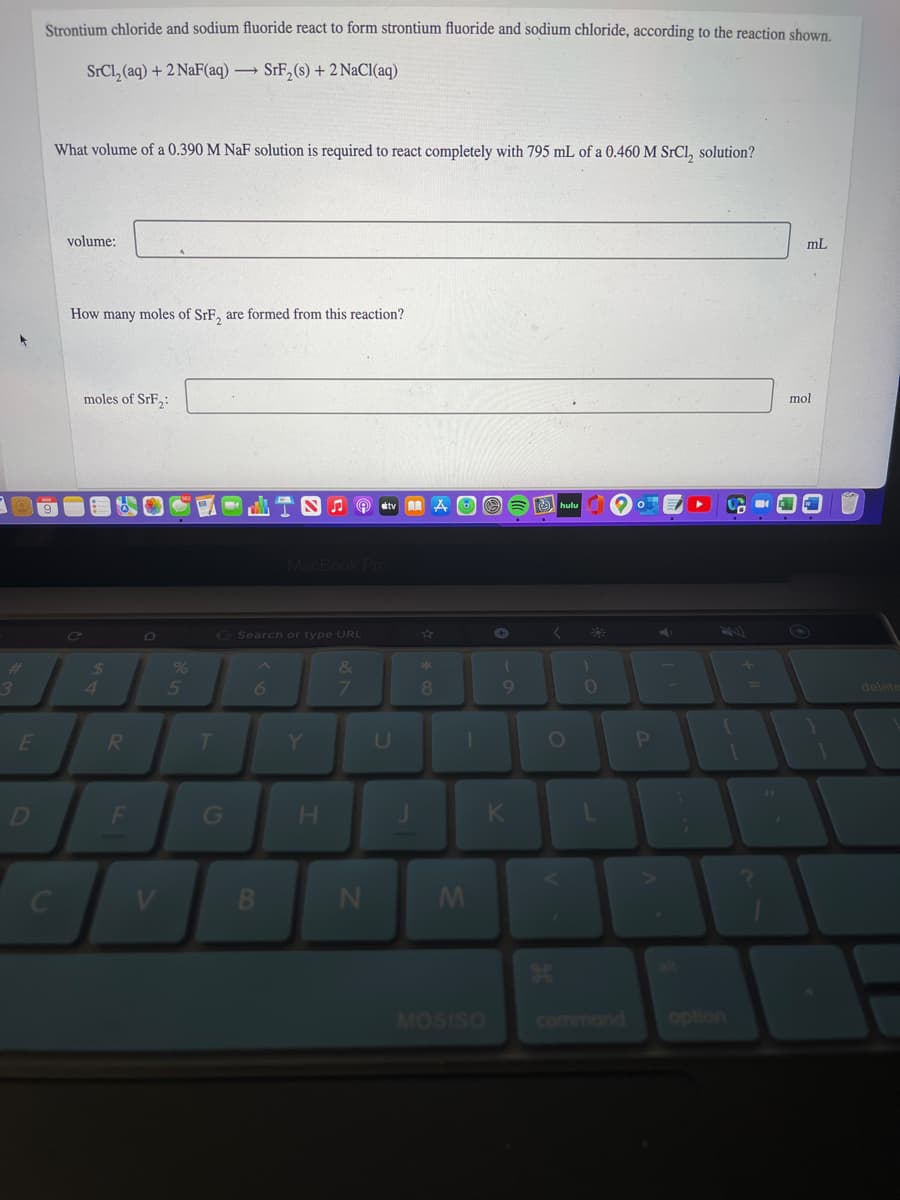
Transcribed Image Text:Strontium chloride and sodium fluoride react to form strontium fluoride and sodium chloride, according to the reaction shown.
SrCl, (aq) + 2 NaF(aq) → SrF,(s) + 2 NaCl(aq)
What volume of a 0.390 M NaF solution is required to react completely with 795 mL of a 0.460 M SrCl, solution?
volume:
mL
How many moles of SrE, are formed from this reaction?
moles of SrF,:
mol
stv
O O 6 hulu
MacBook Pro
G Search or type URL
%
&
3.
5
6
8.
101
delete
E
RI
T.
Y.
D
G
H.
V
MOSISO
command
option
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning