Q: If the bicarbonate ion (HCO3-) concentration in a sample of blood is 0.135 M, determine the carbonic…
A:
Q: Aqueous sulfuric acid (H₂SO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A: Given Mass of H2SO4 = 3.9 g Mass of NaOH = 1.8 g Theoretical yield of Na2SO4= ?
Q: Show all intermediates, mechanisms, and reagents. With each synthetic step in a true synthesis, you…
A: Given tranformation involves C-C bond formation for the synthesis of secondary alcohol. Grignard…
Q: Chemistry a) Which spectrum is more likely to be that of a d'metal ion and why? b) Is the colour of…
A:
Q: Part A If 15.0 mL of glacial acetic acid (pure HC2H3O2; K₂ = 1.8 × 10−5) is diluted to 1.70 L with…
A:
Q: A chemist titrates 90.0 mL of a 0.7050M methylamine (CH3NH₂) solution with 0.5016M HBr solution at…
A:
Q: 4. The following data were obtained for the follwing chemical reaction: 2A+38->2C+D Trial # Initial…
A:
Q: Starting with a structural fragment, draw a complete structural formula by adding all of the…
A: Structural formula generally represent the bonds between participating atoms in a molecule. Here two…
Q: What volume of 0.220 M HBr solution (in mL) is required to obtain 0.0600 moles of HBr? A) 3.67 L B)…
A:
Q: Calculate the change in energy for the following processes and identify if the process is…
A:
Q: For each reaction below, write the conventional, ionic, and net ionic equation. Include designation…
A: Given, Zinc chloride solution is poured into a solution of ammonium carbonate, the net ionic…
Q: For Questions #6 through #14, perform all of the following for each question: 1. calculate the…
A:
Q: Anaesthetic is a used during surgery to cause unconsciousness or a lack of feeling in a part of the…
A: we need to calculate the concentration of the solution in V/V %
Q: On the sketch indicate a. the signs of the electrodes Mg(s) + 2Ag+ (aq) → Mg+2 (aq) + 2Ag(s)
A: Recall the given redox reaction, Mg s + 2 Ag+ aq → Mg+2 aq + 2 Ag…
Q: If 2.56 g of CuNO3 is dissolved in water to make a 0.590 M solution, what is the volume of the…
A: Molarity of solution is defined as numbers of moles of solute dissolved per litre of solution.
Q: What is the product of the following reaction? [1] NK+ CH₂CH₂Br [2] OH/H₂O
A: 1 degree amine will be form ethylamine will be the correct option.
Q: What is the reaction? cycloaddition B HPh 8-8 ∙C- & CO₂Me CO₂Me CO₂Me CO₂Me HPh D [Single selection]…
A:
Q: onsider the following half-reactions: (1) The strongest oxidizing agent is: (2) The weakest…
A:
Q: the "draw structure" buttor the structure of a compounc ster of molecular formula C
A: Organic compounds can be defined as the compounds that contain carbon and hydrogen atoms in a…
Q: Write the equation for the reaction: when nitrogen gas (N2) reacts with sodium metal (Na), solid…
A: Sodium nitride (Na3N) is formed from the reaction of nitrogen gas with sodium metal. Here,…
Q: 34: Draw the molecular diagram of given compounds and predict the bond order and comment on whether…
A: see below
Q: Various ways to measure and represent concentration are available. The following lists the common…
A: Here we have to determine the correct concentration terms used in the following cases.
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A:
Q: Hydrogen gas reduces NO to N₂ in the following reaction: 2H₂(g) + 2NO(g) — 2H₂O(g) + N₂(g) The…
A: Given :- The given reaction is 2H2(g) + 2NO(g) ⇌ 2H2O(g) + N2(g) Temperature = T = 1500 oC
Q: 4. If a given sample of silver and fluorine ideally combine to form 0.6mol of AgF, what is the…
A:
Q: You have 0.685 g of an unknown acid, H₂A, which reacts with NaOH according to the balanced equation…
A:
Q: Discuss the effect of the concentration of the following electrolytes on the conductivity of a…
A: Conductivity of electrolyte depend on the number of ion per unit volume . If the number of ion…
Q: You have a solution of the following ion: [Cr(NH3)6]³+ a) Is the ion more likely paramagnetic or…
A: The nature of diamagnetic and paramagnetic depends on the free ion present in the orbital. If there…
Q: To measure the amount of calcium carbonate (CaCO3) in a seashell, an analytical chemist crushes a…
A:
Q: Calculate the amounts of reagents needed for the preparation of 1.5 L of 0.75 M phosphate buffer, pH…
A: Given: Volume of phosphate buffer solution (V1) = 1.5 L Concentration of phosphate buffer solution…
Q: For each reaction below, write the conventional, ionic, and net ionic equation. Include designation…
A:
Q: Research mass spectrometry and spectrophotometers. How has the use of mass spectrometry advanced our…
A: Mass spectroscopy has an impeccable importance in the real world. Few are listed as follows-
Q: A formula for anti-fungal shampoo contains 2.0% W/V ketoconazole. How many litres (L) of shampoo…
A:
Q: In which of the following scenarios is no change in the internal energy of the system possible? O q…
A:
Q: Identify the INCORRECT statement below Select one: O a. The reaction mechanism is a step-by-step…
A: Many reactions occur in more than one elementary step. A reaction mechanism is a step-by-step…
Q: Complete the following table, which lists information about the measured acid dissociation constants…
A:
Q: Consider the reaction. H CH3 C=C + H H Add curved arrows for the first step. CH₂ H H H-Br: ↑ H :Br:…
A:
Q: Student Z completes his/her calibration step for part 3 and finds an average mass, to be 95.237 g…
A: As per the guidelines, I am allowed to answer one question at a time. Please re upload the rest.…
Q: Which of the ff. statements about the equilibrium constant, Ke is TRUE? 1. Large values mean that…
A: Eqilibrium constant is defined ratio of concentration of products to the concentration of reactant.
Q: Rank the following compounds in order of their basicity, from most to least basic: NH₂ most least…
A: -> Conjugate base of most acidic compound is least basic . ->Carboxylic acid is more acidic…
Q: D E HNO3 H₂SO4 СО2 H+ F ОН E A [H] 1. Fe, HCI 2. OH -СН3 COOH B OH OH NaNO2 HCI C H2O D
A: The synthesis of aspirin can be achieved by nitration of benzene then it's reduction to give aniline…
Q: The reaction Na₂SO4 10H₂O → Na₂SO4(s) + 10H₂O(g) has a K₂ = 5.38 × 10-¹2 at 21.3°C What is its Ke? R…
A:
Q: Gaseous ethane (CH₂CH3) reacts with gaseous oxygen gas (0₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: The IUPAC name for the compound shown is CH₂-CH CH3-CH,C-NH-CH3 NH CHO O N-methylpropionamide…
A: As you post multiple question,we are supposed to answer only one question-first question.kindly…
Q: 20) Which molecule is a primary amide? A) CH,CH2NH(CH3)2 B) CH3NHCOCH3 C) C6H5CONH2 D) CH3NHCH₂CH3
A: Dear student , since you have posted multiple questions we will allow to solve only first question…
Q: 1. Calculate the normality of potassium permanganate (MW: 158.034 g/mol) if 0.2142 g of sodium…
A: Dear student, since the given question is multiple question hence I solved first question according…
Q: Consider the reaction: 2 NaBH4(aq) + H₂SO4(aq) → 2 H₂(g) + Na₂SO4(aq) + B₂H6(9) What volume, in mL,…
A:
Q: Muscone synthesis is possible via an intramolecular Prins reaction. The starting point is…
A:
Q: Click the "draw structure" button to launch the drawing utility. Draw the structure of a molecule…
A:
Q: For Questions #6 through #14, perform all of the following for each question: 1. calculate the…
A: Dear student, As you have asked multiple question but according to guidelines we will solve first…
Step by step
Solved in 2 steps

- Name the structure. Heating compound A in the presence of H2SO4 results in which product?Provide reagents to synthesize sebacoyl chloride from cyclodecane, shownbelow.Compound A has the molecular formula C14H25Br and was obtained by thereaction of sodium acetylide with 1,12-dibromododecane. On treatment ofcompound A with sodium amide, compound B (C14H24) was obtained. Ozonolysisof compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation ofcompound B over Lindlar palladium gave compound C (C14H26), andhydrogenation over platinum gave compound D (C14H28). Further, C yieldedO=CH(CH2)12CH=O on ozonolysis. Assign structures to compounds A through Dso as to be consistent with the observed transformations.
- Give the structures of the two substrates that arerequired to synthesize the following drug againstschizophrenia using the Knorr pyrrole synthesis.tert-Butoxycarbonyl azide was developed as a reagent for peptide synthesis at OWL's home institution, the University of Massachusetts, by Prof. L.A. Carpino. It is prepared by treating tert-butoxycarbonyl chloride with sodium azide. Propose a structure for the initially-formed intermediate in this reaction.When pyrrole is added to a dilute solution of D2SO4 in D2O, 2-deuteriopyrrole is formed. Propose a mecha-nism to account for the formation of this compound.
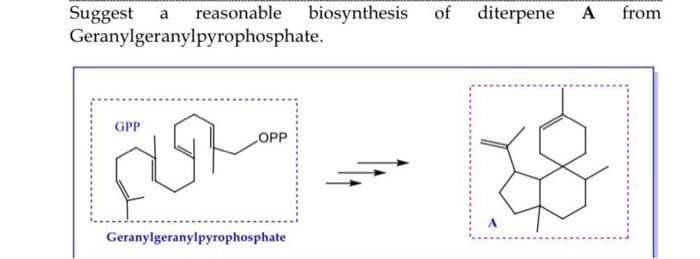
- Choose the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: 1. The molecular formula of compound A is C6H12O6.2. Compound A is a reducing sugar.3. When compound A is subjected to a Wohl degradation two times sequentially, D-erythrose is obtained.4. Compound A is epimeric with D-glucose at C3.5. The configuration at C2 is R.provide a mechanism for the the p450 oxidation of compound 1 below to the corresponding phenol. pls answer asap. Also explain in few sentencessuggest a synthesis of this product from Geranylgeranylpyrophosphate.. and what is the name of the proudct .. Thank you Please hand written.
- Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the stereochemistry drawn. Why isn’t the isomeric epoxide B formed to any extent?Suggest appropriate reagents for the following syntheses.A compound with the formula C8H12 absorbs two equivalents of H2 on Catalina reduction over a palladium catalyst to give compound A. Ozonalysis oxidative cleavage of the unknown compounds gives two compounds B and C.

