Q: 7. Draw the structure for each compound. ( A. acetylene B 3-methyl-1-hexyne C. 4-methyl-2-hexyne D.…
A:
Q: The last step in the synthesis of divinyl ether (used as an anesthetic under the name Vinethene)…
A: In this question, we will prepared the necessary starting material from ethylene which is used in…
Q: For the aqueous [Ni(NH₂)]²* complex K₁ = 5.50 × 108 at 25 °C. Suppose equal volumes of 0.0078M…
A: We need to calculate concentration of Ni2+ at equilibrium
Q: • Which is the most predominant form(s) of Cysteine at the point A on the titration curve? [Select]…
A:
Q: HO-CH₂ H 1. OH 2. CH3(CH2)22 H₂N 3. H O H H CH₂OH H NH₂ O(CH₂)17CH3 H O C-N-C C H -0 4. 0 5. сно HIC…
A: Biomolecules can be defined as the compounds that are present in Living beings.
Q: 1. Question : Arrange the following compounds in increasing order of their boiling points, CH3CHO,…
A: Dear student , since you have posted multiple questions we will allow to solve only first question…
Q: the given below. Wie 1. _H₂ + 0₂ 2. Zn _HCI 3. _Al _Fe(NO3)2 4. _NaNO3 _NaNO₂ NaOH + KNO3 >> 5. _H₂O…
A: The chemical equation is a symbolic representation of the Reaction takes place where one or more…
Q: Question 2 Inversely related to the molecular weight of the sample lipid Saponification number O…
A: Lipids can be defined as the molecules that are made up of hydrocarbon skeleton. They are insoluble…
Q: A 0.2500-g of dry CaCO3 was completely dissolved into a solution of 5-mL conc. HCl and 50-mL…
A:
Q: What is the difference between computational calculations based on Molecular Mechanics and…
A: Computational chemistry is a branch of chemistry that uses computers to assist in solving chemical…
Q: How can you tell the difference between flaky and puff pastry?
A: Pastry have different texture and smoothness which depend upon their making methods .Puff pastry…
Q: With the help of a suitable enthalpy cycle explain what happens when an ionic compound such as NaCl…
A: When NaCL dissolved in water there exists ion-ion dipole bond,which cause stability.
Q: Write the basic equilibrium equation for NH3 NH3(aq) + H₂O(1) NH OH(aq)
A: we have to write basic equilibrium equation for NH3
Q: Fluoridation is the process of adding fluorine compounds to the drinking water. Fluoridated water…
A:
Q: LOH to of H removal of H₂O
A:
Q: Chemistry What is the energy change when fructose is phosphorylated (3.8kcal/mol) and ATP is…
A:
Q: 5-60 SH SH . Which is the most predominant form(s) of Cysteine at the point A on the titration…
A:
Q: 14. The pH of grapefruit juice is 4, what is the pOH?-18X3 1x1044 1x104 15. What is the…
A: Given, The pH of the grapefruit juice is 4, The pOH of the solution is:
Q: 9. What is the volume of 120. grams of neon gas at STP (2-step conversion)?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: C. Draw the skeletal structure and give the IUPAC name for each compound. Condensed Structure…
A:
Q: Match the compound below with its corresponding geometric isomerism identifier
A:
Q: are the tells the story of a chemical reaction. are the new substances that are formed. The large…
A: The answer for all the fill up is shown below.
Q: 2. Some plant fertilizers contain the following compounds: ammonium sulfate, (NH4)₂SO4; calcium…
A: 2. An ionic bond is formed when there is a transfer of electron between bonded atoms. It is formed…
Q: A spherical object has a specific gravity of 4.05 and a radius of 1.16 cm. After this object is…
A:
Q: Consider the electrolysis of an aqueous solution which is 1.0 M NaCl and 1.0 M NiCl₂. Predict the…
A:
Q: If C12H24 reacts with HBr in addition reaction, what is the molecular formula of the product?
A:
Q: 5. Draw the structure of guanine and cytosine correctly indicating the hydrogen onding between them.
A:
Q: give chemical examples of The law of Conservation of Energy and Hess's Law
A: The explanation is given below-
Q: Determine which will produce a precipitate when it reacts with Tollen's reagent a. cyclobutane b.…
A: Tollens’ test, also known as silver-mirror test, is a qualitative laboratory test used to…
Q: Label the conjugate acid-base pairs in each equation: 1. HI + NH3 NH4+ + I- 2. HPO4^2- + OH- PO4^3-…
A:
Q: acid base K K₂b name formula name formula 10 nitrous acid HNO₂ 4.5 x 10 aniline |C HạNH, | 4.3 × 10…
A: Ka value of acetic acid = 1.8×10-5 Ka value of HNO2 = 4.5×10-4 Kb value of aniline = 4.3×10-10…
Q: ! CH₂CH₂ I CH₂CH₂ NAME: CONDENSED FORMULA EXPANDED CONDENSED FORMULA MOLECULAR FORMULA NAME:…
A: Condensed structural formula represents the hydrogen atoms right next to the carbon atoms to which…
Q: What will be the two products after Edman degradation of this dipeptide. (hint: treat with Ph-N=C=S,…
A: In this question, we will draw the structure of the two products after Edman degradation of the…
Q: What happens when water is ejected/expulsion from a gel? Explain
A:
Q: 3. Using the given Kavdues, cdculate the pH of abuffer that contains the given concentrations of a…
A:
Q: Given the following table of standard reduction potentials for a hypothetical element X under an…
A: Note- Dear student it is a mutipart questions, hence I solved first three questions according to…
Q: Determine how many grams of CO2 13.72 g carbon dioxide are produced by burning 5.45 g of C4H10.
A:
Q: Draw the structure(s) of the major organic product(s) of the following reaction. -N₂ HSO4 CuCN KCN…
A: Aromatic amine reacts with sodium nitrite and hydrochloric acid at 0oC temperature to form a…
Q: is drawn below (left). Complete 10. One cis, trans isomer of 3,5-dimethylcyclohexanol the drawings…
A: The stability of cyclohexane is determined by the position of the substituent on the cyclohexane…
Q: 2 (3) Expected IK H HO Но 500 peaks pn 201 shew c bios H
A: IR tells us about the functional groups present in the compound.
Q: (atoms) Number of Spheres Per Unit Cell: Volume of Spheres Per Unit Cell: π ³. Number of sphere per…
A: There are mainly following type of unit cells , which have different numbers of atoms/Molecules and…
Q: 4. A sample of the amino acid cystine has a specific rotation of -68°. Pure (-)-cystine has a…
A:
Q: Which of the following does not contribute to the stability of benzene? a. overlapping pi electrons…
A: Highly stable conjugated cyclic compounds that do not give addition reactions and follow the Huckel…
Q: OOH О-С-ОН 0 С-ОН 0 С-ОН Br NO₂ H2C-CH3 ОН . V II IV Rank the following acids by acid strength, from…
A: ->For Acidity we see stability of conjugate base if conjugate base is more stable then it is most…
Q: Convert the experimental glucose concentration (provided in the table below) from mM (millimolar) to…
A:
Q: Write the condensed structures corresponding to the following names. 1. 3-ethyl-5; 5-dimethylectanal…
A:
Q: How many milliliters of lactic acid must be added to 1L of water to produce a solution of 0.477 mM?…
A: The correct answer is given below
Q: Ni(CO).(g) ← 2 03(g) → 3 O₂(g) Ni(s) + 4 CO(g)
A: Write the expression for Kc (equilibrium constant) for the following reactions.
Q: 6. Draw four alternative, reasonable resonance structures to represent the anion show below. Also,…
A: It is the path to describe the bonding in a certain molecule by combination of certain contributing…
Q: Alex’s mass before getting the tattoo was 61.5 kg. The tattoo contains 5.20x1023 molecules of iron…
A: Given, Alex’s mass before the tattoo = 61.5 kg tattoo contains 5.20x1023 molecules of Fe3O4
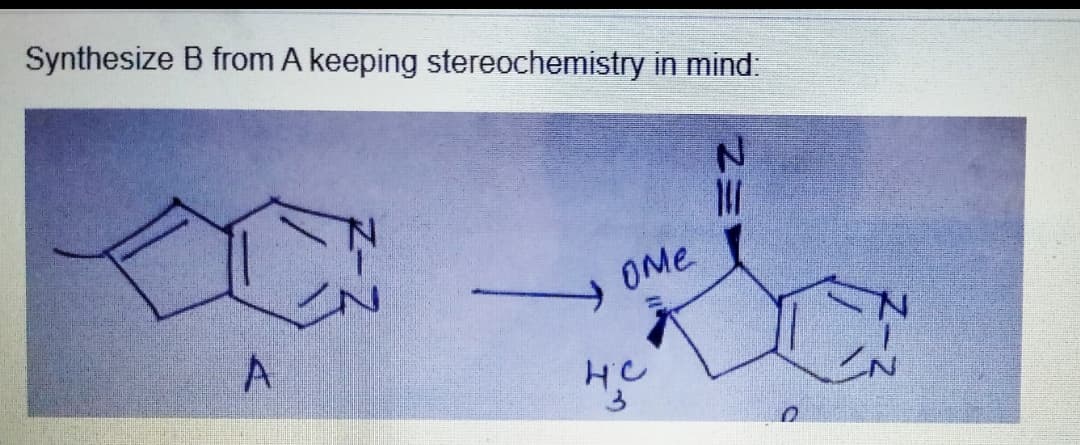
Step by step
Solved in 2 steps with 1 images

- Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.Draw the products of these reactions clearly showing stereochemistry in each case if necessary. If the product is formed as a racemic mixture, clearly draw one stereoisomer with dash/wedge bonds and write “+en”. Circle whether each reaction occurs by an SN1 or SN2 pathway.Identify the missing reagents a-f in the following scheme:
- Identify the reagents a-c in the following scheme:a) Draw the mechanism, using good curved arrow notation, for the reaction of 2-methyl-2- butene with bromine in water, omitting any stereochemistry: b) (R)-3-butyn-2-ol is treated with 2 mol of HBr, yielding optically active Compound A (C4H8OBr2). Draw the structure of the reaction with the correct stereochemistry. c) Name Compound A above, including stereochemistry. d) Compound A is treated with HBr to yield Compound B (C4H7Br3). The resulting solution is optically inactive. Draw the product(s) obtained and explain why there is a loss in optical activity.Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related?
- Which compound would undergo carbocation rearrangement during an SN1 reaction? A B C DElimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain whyName attached compound and decide which stereoisomer will react faster inan E2 elimination reaction. Explain your choice.
- Draw the products for the following chemical reactions, indicating stereochemistry wherever applicable. Indicate if the reaction proceeds via SN1 or SN2 mechanismWhen the following stereoisomer of 2-bromo-1,3-dimethylcyclohexane is treated withsodium methoxide, no E2 reaction is observed. Explain why this compound cannotundergo the E2 reaction in the chair conformation.Draw the product formed (including stereochemistry) in attached pericyclicreaction.

