Table 2. Data on Sample Analysis Volume of water sample (mL) : 10.00mL Trial Volume of EDTA (mL) ppm CaCO3 1 0.19 0.16 3 0.19 Average ppm CaCO3
Q: emical apparatus te easure potassium?
A: Answer: to measure the potassium the method are used atomic absorption or inductivity coupled…
Q: The reaction between nitrogen monoxide and oxygen is given below: 2 NO(g) + 02(9) - 2 NO2(g) We…
A:
Q: 2. Identity the reagent sequence in the following transformations (retrosynthesis). A. OH B. он
A: These conversions can be done with the help of grignard Reagents and substitution reaction.
Q: A 50.0 mL HCI is pipetted out from a 1000-mL 0.0250 M stock solution. To this 50.0mL aliquot, a…
A: Given-> Volume of HCl pipette out = 50.0 ml Volume of stock solution = 1000 ml Molarity of stock…
Q: What is the mass in grams of precipitate formed when 250.0 mL of 0.400 M ZnCI, is mixed with excess…
A: Given-> Volume of ZnCl2 = 250.0 ml Molarity of ZnCl2 = 0.400 M
Q: Assign oxidation number of the manganese in K2MNO4: а. +6 O b. +2 О с. +4 O d. +7
A:
Q: Illustrate the carbocation pathway for the following reactions + HCI + CH3-OH
A: In carbocation pathway reaction proceeds through formation of carbocation and the carbocation must…
Q: Salicylic acid is added into a test tube, followed by methanol and sulfuric acid as the catalyst. It…
A: Given that, salicylic acid is added into a test tube, followed by methanol and sulfuric acid as the…
Q: suged to rearangenert 8) Dehydration of an unknown alcohol with concentrate@ H2SO4 results in the…
A: Note : Alcohols undergo acid catalyzed rearrangement. ( see below).
Q: in mind that HCN is a weak acid. acids: O bases: O ? other: O acids: O bases: O O other: O
A: Acid base reaction takes +place between KOH and HCN. HCN + KOH --> KCN + H2O As both have same…
Q: Br Br > WHat kind oF (caalon is this? E2IE|SNI|SN2 1, MgBr 2. H20 Ho heat CH3 CH21 AIC13
A: Note : carbonyl compounds undergo nucleophilic addition reactions. 1,5-hexadiene undergo cope…
Q: II. GENERAL INSTRUCTIONS Read and analyze carefully each problem. Determine the (a) rate law, (b)…
A: 2A +2B +2C ------> products Rate law for the above reaction Rate = k [A]m[B]n[C]p Where m, n,…
Q: Need to find percent recovery of crude caffine and purified caffine
A: Given that - Mass of Caffeine in tea bags = 550 mg Mass of Crude Caffeine = 109 mg Mass of…
Q: Hint: you need to loose a carbon from one of the starting materials. Insctie pts) OCH3 Hint: you…
A: Note : Retrosynthesis followed by synthesis.
Q: 10. Alkyl diazonium salts decompose to form carbocations, which go on to form products of…
A:
Q: create three (3) multiple-choice questions on either Precipitation titration, redox titration, or…
A: A redox titration is a titration process in which the analyte and titrant react through an oxidation…
Q: An oil boils at a lower temperature when steam is added to it because: O the water dissolves in the…
A: We have to find the reason for why the boiling point decreases when steam is added to oil.
Q: A compound with molecular formula C3H100 has the H NMR spectrum shown. Which of the choices shows…
A: C8H10O ( DBE = 8- (10/2) + 1 = 9-5 = 4 . The compound name is ethoxy benzene.
Q: Look up the Born-Haber Cycle and read about it (book or internet). Use the following information and…
A: Given-> Na(s) ---> Na(g). ∆H° = 109 kJ Cl2(g) ---> 2Cl(g). ∆H° = 243 KJ Na(s)…
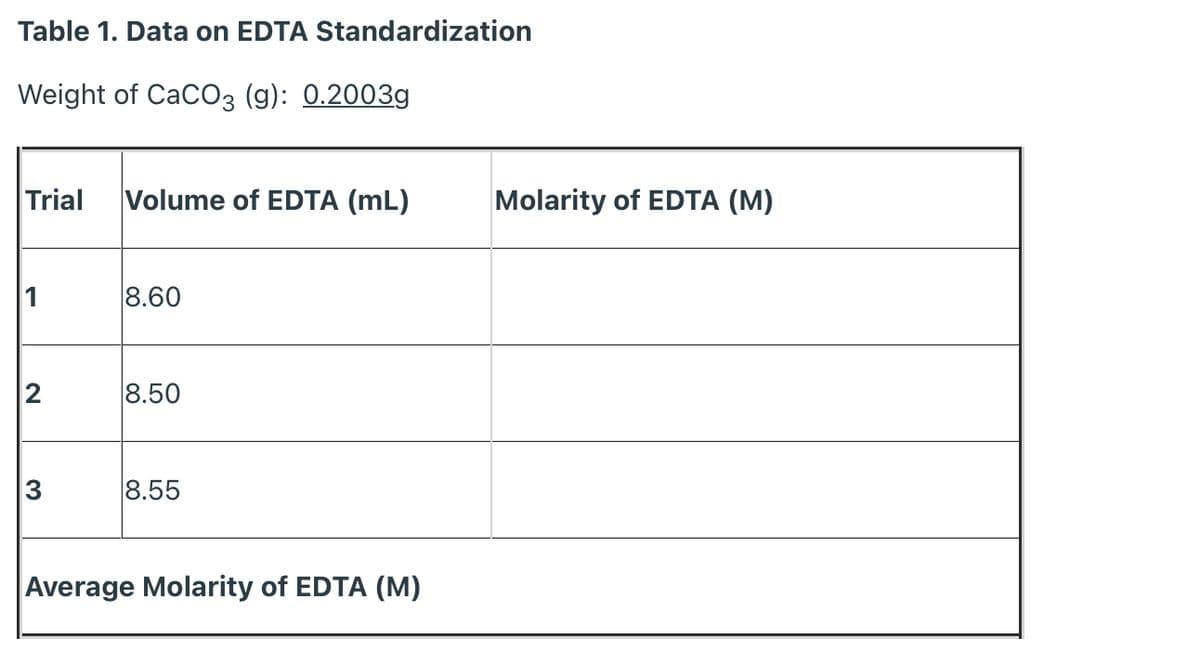
Q: Weight of CaCO3 (g): 0.2003g Trial Volume of EDTA (mL) Molarity of EDTA (M) 1 8.60 2 8.50 8.55…
A: 1 mol EDTA neutralises 1 mol of CaCO3
Q: On the basis of the 18-electron rule, identify the first-row transition metal for each of the…
A:
Q: (ve
A: The resistance defines the hindrance to the mobility of the ions in a solution and it can be…
Q: Chemistry Calculate the ΔE°' for the following reaction: ascorbate + FAD → dehydroascorbate +…
A: Here we are required to find the cell potential of the given electrochemical reaction ascorbate +…
Q: (g (n' (h M(n-CSH5):(CO)(M=M bond) 3 Calculate the Total Valence Electron (TVE) and sketch the…
A: Total valence electrons of a complex is equals to the valence electron of metal (d + s electrons)…
Q: Draw a voltaic cell of the following reaction below (please include anode, cathode, and flow of…
A: This question is related to electrochemical cells. Voltaic cell is an electrochemical cell in which…
Q: Question 8 Listen -LENGTH- -GAP A 11.4 inch long aluminum alloy bar is placed between two rigid,…
A:
Q: ball & stick + labels The molecule has a carbonyl group. A general functional group representation…
A: Given, Based on ball and stick model the structure of the molecule is:
Q: One of the most promising possibilities for commercial fusion is combining deuterium (amu 2.014102…
A: Here we are required to find the energy that would be given off by nuclear reaction
Q: The carbon−14 decay rate of a sample obtained from a young tree is 0.250 disintegration per second…
A:
Q: List the following from lowest to highest first ionization energy. Cl, Na, K
A: Ionization energy : The energy required to remove the highest energy electron i.e.valence electron…
Q: What element is being oxidized in the following redox reaction? C10H22O2(aq) + KMnO4(aq)…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: For the reaction below, Kc = 1.10 × 10⁻⁸. Note Kc is sometimes called K. What is the equilibrium…
A:
Q: how many moles of Ca3(PO4)2will be formed if 8.0 mole of each reactant is present?…
A:
Q: heat (in joules
A:
Q: 24-26. Predict the product of the reaction sequence shown below. 1.KMNO,KOH; 2. H H,O(excess) H,SO,…
A: Gabriel phthalimide synthesis: In the Gabriel synthesis an alkyl halide is converted into a primary…
Q: One of the buffers that can be used for running an agarose gel is 1x TBE (tris borate EDTA) buffer.…
A: On dilution concentration changes but no of moles remain same. So, M1V1 = M2V2 M1: Concentration of…
Q: Which of the following structures would be most readily reduced by a hydride delivery agent, such as…
A: ->NaBH4 and LiAlH4 are reducing agent . These are reducing agent because produce H- nucleophile .…
Q: The pKa's of H3PO4 are 2.0, 7.0, and 12.5. Assuming you have the sodium salts of all of the ionic…
A: Here we have to find concentration proton donor and proton acceptor respectively in 0.1 M phosphate…
Q: Which of the following metals will NOT dissolve in nitric acid or hydrochloric acid? Ca Mg K Fe…
A:
Q: Given the following unbalanced chemical reaction, what volume of 0.650 mol/L K2CrO4 is required to…
A: Balanced equation is 2 AgNO3 + K2CrO4 --> Ag2CrO4 + 2 KNO3 Use the formula M1V1 /n1 = M2…
Q: The reaction between nitrogen monoxide and oxygen is given below: 2 NO(g) + 02(9) 2 NO2(9) We…
A:
Q: The combustion of isoctane reaction produces -5099.5 kJ of heat per mole of C8H18 (g) consumed,…
A: Given data , □Hcomb = -5099.5 kJ of heat per mole of C8H18 (g) (□Hf) for CO2(g) = -393.5 kJ/mol…
Q: An Arrhenius plot (ln k vs. 1/T) of data gathered while measuring the effect of temperature on the…
A: Given that the equation of trend line, y=−6481.3x+14.945 we have to find activation energy Ea.
Q: Consider the reaction below. If you start with 7.00 moles of C3Hg (propane) and 7.00 moles of 02,…
A:
Q: OH- + CH3COOH ==== CH3COO- + H2O . Which of the products is a conjugate base?
A: Given :- OH- + CH3COOH → CH3COO- + H2O To identify :- Conjugate base
Q: What products are formed when each of the following amines is treated with HCI: (a) CH,CH,NH,:
A: Given; reactants
Q: Balance the following equation with the smallest whole number coefficients. Choose the answer that…
A:
Q: In each pair of isotopes shown, indicate which one you would expect to be radioactive. 20 17 40 45…
A: Radioactivity can be defined as the process in which there is spontaneous emission of radiation in…
Q: HCO3- + HCl ==== H2CO3 + Cl- . Which one of reactants is an acid?
A:
Q: The Kp for the reaction A (g) ⇌ 2 B (g) is 0.0410. What is Kp for the reaction 2 B (g) ⇌ A (g)?
A:

Step by step
Solved in 2 steps with 2 images

- The thickness of the chromium plate on an auto fender is determined by dissolving a 30.0-cm2 section in acid and oxidizing Cr3+ to Cr2O7 2-with peroxydisulfate. After removing excess peroxydisulfate by boiling, 500.0 mg of Fe(NH4 )2 (SO4 )2•6H2O is added, reducing the Cr2O7 2- to Cr3+ . The excess Fe2+ is back titrated, requiring 18.29 mL of 0.00389 M K2Cr2O7 to reach the end point. Determine the average thickness of the chromium plate given that the density of Cr is 7.20 g/cm3 . Note: thickness = volume /areaCalculate the amount of manganese (54.94 g/mol) from a processed ore using the following data. Standardization data: 2.000 mL KMnO4 = 0.02015 g Na2C2O4 (134 g/mol) Sample analysis data: Weight of sample = 0.1012 g Na2C2O4 used(in excess)= 0.3200 g KMnO4 used for back-titration = 9.93 mL Rxn: 2MnO4- + 5C2O42- + 16H+ → 2Mn2+ + 10CO2 + 8H2Owhat wt. of limestone containing 9.57% Mg must be taken for analysis in order to precipitate of 0.551g Mg2P2O7? how many grams of Na2SO4 are required to ppte Ag2SO4 from 2.000t of AgNO3? a sample of magnetite (impure Fe3O4) weighing 0.5000g is fused with oxidizing flux and the ferric compound formed is eventually precipitated as ferric hydroxide and ignited to ferric oxide which weighs 0.4980calculate %Fe & %Fe2O3
- An exhausted zeolite softener was regenerated by passing 100 litres, of NaCl. Solution containing 150 gm per lit. of NaCl. How many lit. of a sample of H2O of hardness 300 ppm can be softened by this softener? (Given at wts. for C = 12, O = 16, Na = 23, CI = 35.5, Ca = 40).Determine the solubility of KIO4 in water. Volume of saturated KIO4 in conical flask is 25.0 ml,KI is 2g,H2SO4 (3.0M) is 20.0 ml added to KI solution , volume of 0.200 M sodium thiosulphate used for titration is 8.00 ml.and if mean ionic activity co efficient,y+- of KIO4 in the saturated solution is 0.85 ,what is Ksp. .The concentration of a solution of EDTA was determined by standardizing against a solution of Ca2+ prepared from the primary standard CaCO3. A 0.4302g sample of CaCO3 was transferred to a 60mL volumetric flask, dissolved using a minimum of 6 M HCl solution, and diluted to volume. A 44mL portion of this solution was transferred into a 250-mL Erlenmeyer flask and the pH adjusted by adding 5 mL of a pH 10 NH3-NH4 L buffer containing a small amount of Mg2+ EDTA. After adding calmagite as a visual indicator, the solution was titrated with the EDTA, requiring 632mL to reach the end point. Calculate the molar concentration of the titrant
- What is the percentage of Nickel in an ore if, when analyzed by the cyanide method, 20.00mL of KCN solution (containing 1.00mmol of AgNO3 per milliliter) are used? Wt of sample used = 0.2500gwhat mass in grams of Na2S2O3 is needed to dissolve 4.7 g of AgBr in a solution volume of 1.0 L given that Ksp for AgBr is 3.3 x 10^-13 and Kf for [Ag(S2O3)2]^3- is 4.7 x 10^13?What is the concentration of ions in the soil solution after fertilizer application? Suppose that 122 pounds of K+ were applied per acre, then a gentle rain soaked the top 10 inches of soil to field capacity, which for the given soil was about 16% water by volume. If the K+ was applied as KCl, it is plausible that it all dissolved and distributed relatively uniformly with the infiltrating water. If so, then what was the K+ concentration in the soil solution in mol K+/L solution? Note that the volume can be computed like we do for an acre-furrow-slice (AFS), as area times depth. This is going to be a relatively small number, so please report your answer in mol K+/L solution to at least 5 decimal places.
- A piece of an unknown weighing 12.50is placed in 49.7cm of water in a cylinder. The water level increases to 69.0cm3. whats the calculated value for the drnsity of the solid in g ml-1A 25.00 mL aliquot (a portion or part) of the standard Ca (II) solution (prepared by dissolving 0.3139 g CaCO3 in water and diluted to 250 mL) reacts with 27.25 mL of the EDTA solution. What is the molarity of the EDTA? Show calculation below.A 46.4046.40 mL aliquot from a 0.4850.485 L solution that contains 0.3500.350 g of MnSO4MnSO4 (MW=151.00MW=151.00 g/mol) required 35.235.2 mL of an EDTA solution to reach the end point in a titration. What mass, in milligrams, of CaCO3CaCO3 ( MW=100.09 MW=100.09 g/mol) will react with 1.691.69 mL of the EDTA solution?

