The adjusted trial balance for Sheridan Company is given below. SHERIDAN COMPANY Trial Balance August 31, 2022 Before After Adjustment Adjustment Dr. Cr. Dr. Cr. Cash $11,320 $11,320 Accounts Receivable 8,930 9,580 Supplies 2,630 1,330 Prepaid Insurance 4,000 2,640 Equipment 16,000 16,000 Accumulated Depreciation-Equipment $3,600 $4,800 Accounts Payable 5,800 5,800 Salaries and Wages Payable 1,500 Unearned Rent Revenue 2,070 1,140 Common Stock 12,390 12,390 Retained Earnings 5,340 5,340 Dividends 2,750 2,750 Service Revenue 32,590 33,240 Rent Revenue 13,320 14,250 Salaries and Wages Expense 16,870 18,370 Supplies Expense 1,300 Rent Expense 12,610 12,610 Insurance Expense 1,360 Depreciation Expense 1.200 $75,110 $75,110 $78.460 $78,460 Prepare the closing entries for the temporary accounts at August 31. (If no entry is required, select "No Entry" for the account titles and enter O for the amounts. Credit account titles are automatically indented when the amount is entered. Do not indent manually.)
The adjusted trial balance for Sheridan Company is given below. SHERIDAN COMPANY Trial Balance August 31, 2022 Before After Adjustment Adjustment Dr. Cr. Dr. Cr. Cash $11,320 $11,320 Accounts Receivable 8,930 9,580 Supplies 2,630 1,330 Prepaid Insurance 4,000 2,640 Equipment 16,000 16,000 Accumulated Depreciation-Equipment $3,600 $4,800 Accounts Payable 5,800 5,800 Salaries and Wages Payable 1,500 Unearned Rent Revenue 2,070 1,140 Common Stock 12,390 12,390 Retained Earnings 5,340 5,340 Dividends 2,750 2,750 Service Revenue 32,590 33,240 Rent Revenue 13,320 14,250 Salaries and Wages Expense 16,870 18,370 Supplies Expense 1,300 Rent Expense 12,610 12,610 Insurance Expense 1,360 Depreciation Expense 1.200 $75,110 $75,110 $78.460 $78,460 Prepare the closing entries for the temporary accounts at August 31. (If no entry is required, select "No Entry" for the account titles and enter O for the amounts. Credit account titles are automatically indented when the amount is entered. Do not indent manually.)
Chapter4: The Adjustment Process
Section: Chapter Questions
Problem 11EA: Prepare adjusting journal entries, as needed, considering the account balances excerpted from the...
Related questions
Question
100%
see attached image
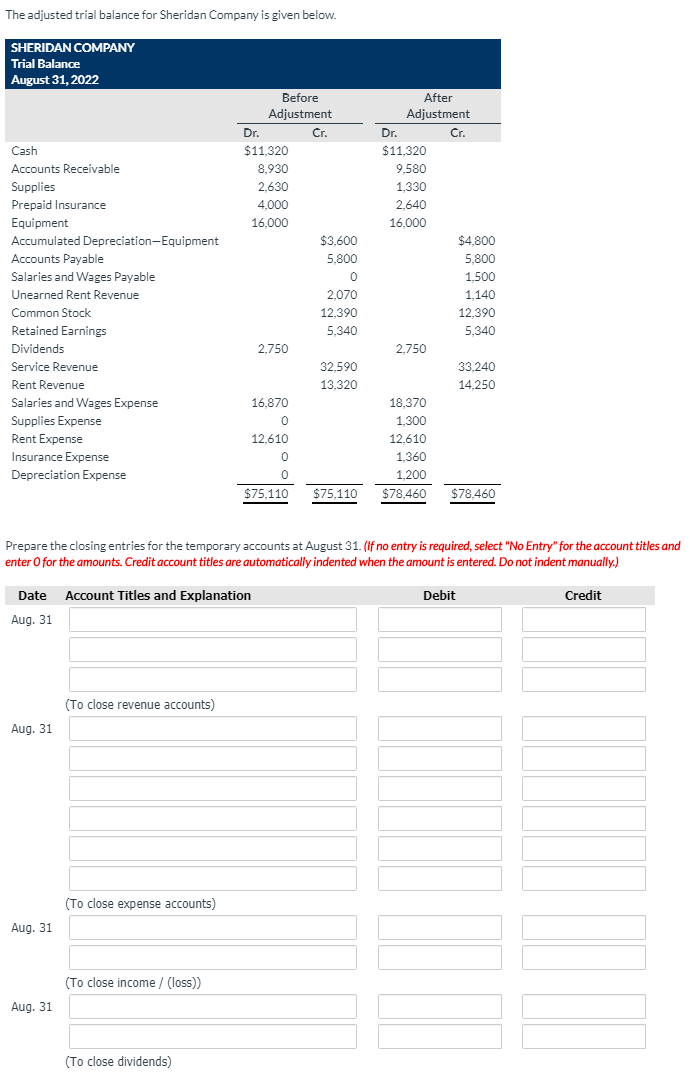
Transcribed Image Text:The adjusted trial balance for Sheridan Company is given below.
SHERIDAN COMPANY
Trial Balance
August 31, 2022
Before
After
Adjustment
Adjustment
Dr.
Cr.
Dr.
Cr.
Cash
$11,320
$11,320
Accounts Receivable
8,930
9,580
Supplies
2,630
1,330
Prepaid Insurance
4,000
2,640
Equipment
16,000
16,000
Accumulated Depreciation-Equipment
$3,600
$4,800
Accounts Payable
5,800
5,800
Salaries and Wages Payable
1,500
Unearned Rent Revenue
2,070
1,140
Common Stock
12,390
12,390
Retained Earnings
5,340
5,340
Dividends
2,750
2,750
Service Revenue
32,590
33,240
Rent Revenue
13,320
14,250
Salaries and Wages Expense
16,870
18,370
Supplies Expense
1,300
Rent Expense
12,610
12,610
Insurance Expense
1,360
Depreciation Expense
1.200
$75,110
$75,110
$78.460
$78,460
Prepare the closing entries for the temporary accounts at August 31. (If no entry is required, select "No Entry" for the account titles and
enter O for the amounts. Credit account titles are automatically indented when the amount is entered. Do not indent manually.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
I need the income statement of the above transactions. Thank you
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, accounting and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Accounting Volume 1
Accounting
ISBN:
9781947172685
Author:
OpenStax
Publisher:
OpenStax College

Survey of Accounting (Accounting I)
Accounting
ISBN:
9781305961883
Author:
Carl Warren
Publisher:
Cengage Learning

Intermediate Accounting: Reporting And Analysis
Accounting
ISBN:
9781337788281
Author:
James M. Wahlen, Jefferson P. Jones, Donald Pagach
Publisher:
Cengage Learning

Principles of Accounting Volume 1
Accounting
ISBN:
9781947172685
Author:
OpenStax
Publisher:
OpenStax College

Survey of Accounting (Accounting I)
Accounting
ISBN:
9781305961883
Author:
Carl Warren
Publisher:
Cengage Learning

Intermediate Accounting: Reporting And Analysis
Accounting
ISBN:
9781337788281
Author:
James M. Wahlen, Jefferson P. Jones, Donald Pagach
Publisher:
Cengage Learning


Cornerstones of Financial Accounting
Accounting
ISBN:
9781337690881
Author:
Jay Rich, Jeff Jones
Publisher:
Cengage Learning
Expert Answers to Latest Homework Questions
Q: 15. Which statement below best describes the difference between an intermediate and a transition…
Q: Need help please
Q: Determine the molar solubility for CaF2 by constructing an ICE table,
writing the solubility…
Q: Calculate the volume of each gas sample at STP.
22.7 mol Cl2
Q: On the graph on right, the constraints C₁ and C₂ have been
plotted.
Using the point drawing tool,…
Q: A gas mixture contains 84% nitrogen and 16% oxygen.
If the total pressure is 1.04 atm, what are the…
Q: Need help please
Q: A heliox deep-sea diving mixture delivers an oxygen partial pressure of 0.25 atm whne the total…
Q: Can you show me how to create a basic HTML page?
Q: Actuary and trustee reports indicate the following changes in the PBO and plan assets of Mahomes…
Q: 4. (25 pts) In an RLC circuit, a power source provides an oscillating emf given by
E = 6.00 sin(42t)…
Q: Give me one solid conclusion paragraph on The Role of the Strategic Management Accounting in the…
Q: On June 13, the board of directors of Siewert Incorporated declared a 5% stock dividend on its 40…
Q: An empty steel container is filled with 3.00 atm
of H2 and 3.00 atm of F2. The system is allowed
to…
Q: Gwinnett Park Co. reported net income of $253,300 for its fiscal year ended September 30, 2017. At…
Q: A gas mixture contais each of the following gases at the indicated partial pressure.
N2 - 219 torr…
Q: An experiment shows that a 258 mL gas sample has a mass of 0.427 g at a pressure of 750 mm Hg and a…
Q: What are the units for the rate constant for a seventh order reaction?
Q: Mutation a change in the DNA
Point mutations - when a single nitrogen base is deleted, inserted or…
Q: Draw out the mechanism please
Q: A cylinder contains 29.0 L of oxygen gas at a pressure of 1.9 atm and a temperature of 289 K.
How…