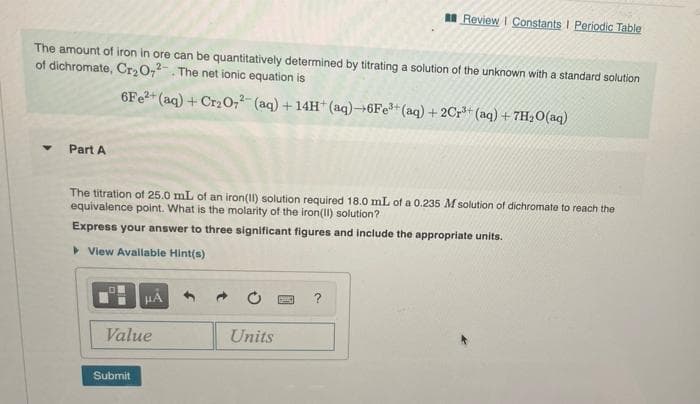
The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown with a standard solution of dichromate, Cr2O,. The net ionic equation is 6FE2* (aq) + Cr20,2 (aq) + 14H*(aq)→6FE**(aq) + 2Cr (aq) + 7H2O(aq) Part A The titration of 25.0 mL of an iron(II) solution required 18.0 mL of a 0.235 M solution of dichromate to reach the equivalence point. What is the molarity of the iron(11) solution? Express your answer to three significant figures and include the appropriate units. > View Avallable Hint(s)
The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown with a standard solution of dichromate, Cr2O,. The net ionic equation is 6FE2* (aq) + Cr20,2 (aq) + 14H*(aq)→6FE**(aq) + 2Cr (aq) + 7H2O(aq) Part A The titration of 25.0 mL of an iron(II) solution required 18.0 mL of a 0.235 M solution of dichromate to reach the equivalence point. What is the molarity of the iron(11) solution? Express your answer to three significant figures and include the appropriate units. > View Avallable Hint(s)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter4: Stoichiometry Of Chemical Reactions
Section: Chapter Questions
Problem 80E: What is the concentration of NaCl in a solution if titration of 15.00 mL of the solution with 0.2503...
Related questions
Question
Transcribed Image Text:I Review I Constants I Periodic Table
The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown with a standard solution
of dichromate, Cr0,. The net ionic equation is
6F22+ (aq) + Cr20,2- (aq) + 14H (aq)→6FE (aq) + 2Cr* (aq) + 7H2O(aq)
Part A
The titration of 25.0 mL of an iron(Il) solution required 18.0 mL of a 0.235 M solution of dichromate to reach the
equivalence point. What is the molarity of the iron(II) solution?
Express your answer to three significant figures and include the appropriate units.
> View Avallable Hint(s)
HẢ
Value
Units
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax