The Claisen condensation converts two molecules of an ester into a ß-keto ester. The reaction starts with the ester in an alkoxide/alcohol solution and is worked up with acid to form the neutral ß-keto ester product. 1. "OCH,CH3 / CH3CH,OH 2 2. H3O* Show the curved-arrow mechanism for the Claisen condensation of ethyl ethanoate treated with ethoxide ion. Include all formal charges and nonbonding electrons. In each step, draw only the species that react in that step. If an enolate resonance form is possible, draw only the carbanionic form. If a CH; group is being deprotonated, draw all H's on the reacting site. Tip: always omit ethanol byproducts.
The Claisen condensation converts two molecules of an ester into a ß-keto ester. The reaction starts with the ester in an alkoxide/alcohol solution and is worked up with acid to form the neutral ß-keto ester product. 1. "OCH,CH3 / CH3CH,OH 2 2. H3O* Show the curved-arrow mechanism for the Claisen condensation of ethyl ethanoate treated with ethoxide ion. Include all formal charges and nonbonding electrons. In each step, draw only the species that react in that step. If an enolate resonance form is possible, draw only the carbanionic form. If a CH; group is being deprotonated, draw all H's on the reacting site. Tip: always omit ethanol byproducts.
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter16: Aldehydes And Ketones
Section: Chapter Questions
Problem 16.26P: Wittig reactions with the following -chloroethers can be used for the synthesis of aldehydes and...
Related questions
Question
Step 5b: Add curved arrows to show formation of the product
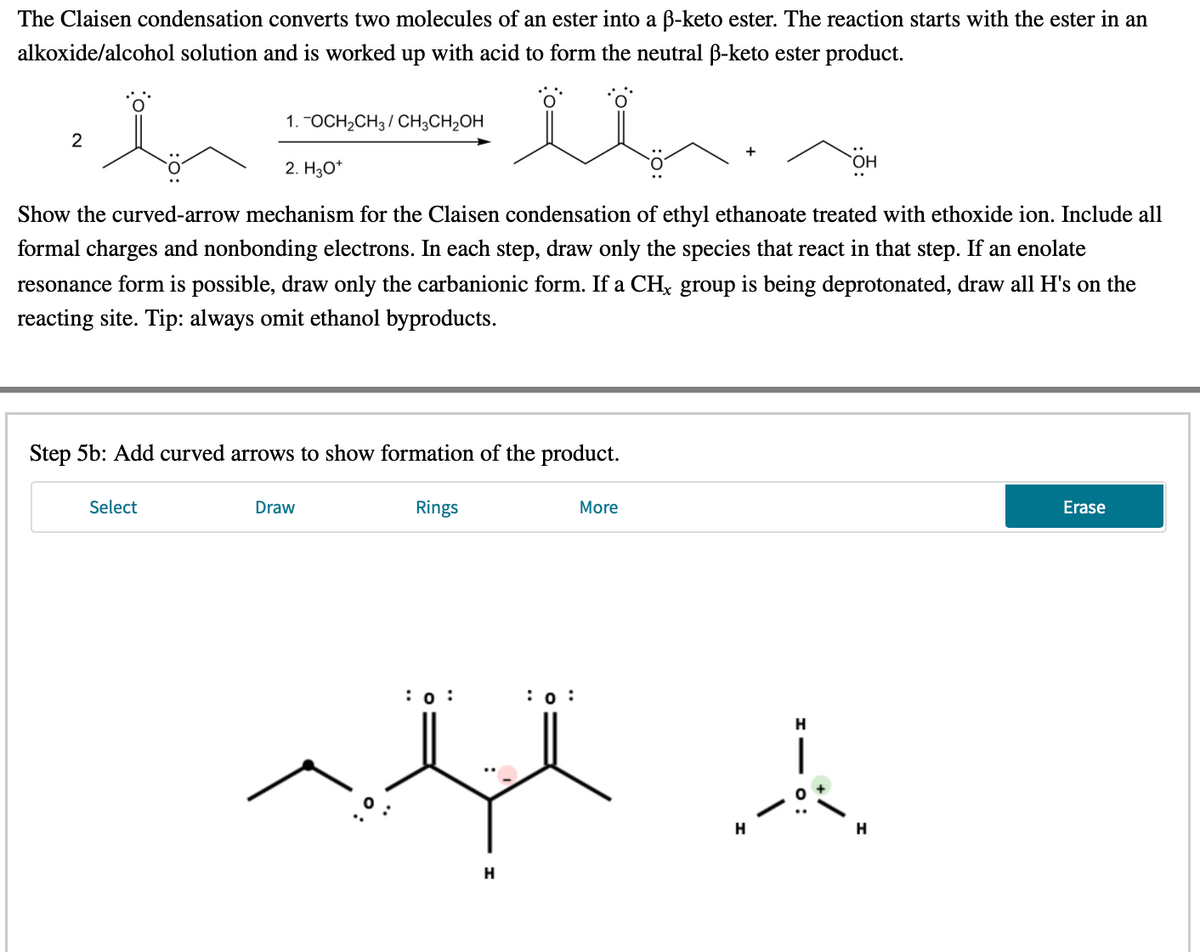
Transcribed Image Text:The Claisen condensation converts two molecules of an ester into a B-keto ester. The reaction starts with the ester in an
alkoxide/alcohol solution and is worked up with acid to form the neutral B-keto ester product.
1. "OCH2CH3/ CH3CH2OH
2. Hо*
Show the curved-arrow mechanism for the Claisen condensation of ethyl ethanoate treated with ethoxide ion. Include all
formal charges and nonbonding electrons. In each step, draw only the species that react in that step. If an enolate
resonance form is possible, draw only the carbanionic form. If a CHx group is being deprotonated, draw all H's on the
reacting site. Tip: always omit ethanol byproducts.
Step 5b: Add curved arrows to show formation of the product.
Select
Draw
Rings
More
Erase
: о :
:0 :
H
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning