Chapter15: Complex Acid/base Systems
Section: Chapter Questions
Problem 15.29QAP
Related questions
Question
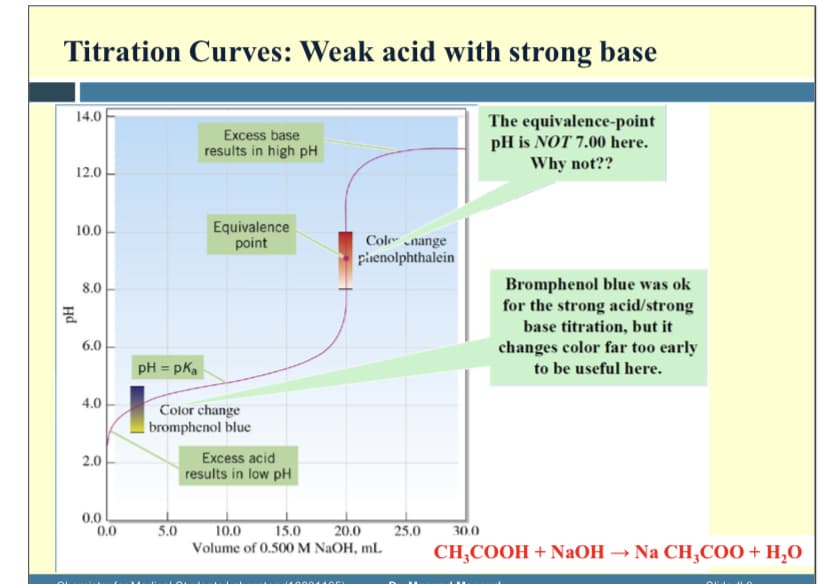
Transcribed Image Text:Titration Curves: Weak acid with strong base
14.0
The equivalence-point
pH is NOT 7.00 here.
Why not??
Excess base
results in high pH
12.0
Equivalence
point
10.0
Cole „nange
piienolphthalein
Bromphenol blue was ok
for the strong acid/strong
base titration, but it
changes color far too early
8.0
6.0
pH = pka
to be useful here.
4.0
Color change
| bromphenol blue
Excess acid
results in low pH
2.0
0.0
0.0
5.0
10.0
15.0
20.0
25.0
30.0
Volume of 0.500 M NaOH, mL
CH;COOH + NaOH → Na CH,CO0 + H,0
Hd
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning



Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning