Q: Draw the products of the two step reaction sequence shown below. Use dash and/or wedge bonds to…
A: In the reaction when the CH3CH2MgBr reagent reacts with the reactant, we get the Alcohol…
Q: The H⁺ concentration in an aqueous solution at 25 °C is 5.7 × 10⁻⁶. What is [OH⁻]?
A: Autoionization of water takes place as follows:In pure water, the concentration of the hydronium ion…
Q: Both silver(I) bromide and silver(I) iodide are slightly soluble salts, with Ksp values at 25°C of…
A:
Q: Re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in…
A: Size increases as we move down the groupSize decreases as move along the period. Size of anion is…
Q: What nucleophile is required to produce this product? Select one: a. NaSH O b. OTS O d. e. t MgBr…
A: Nucleophilic substitution is an important organic reaction in which a leaving group is replaced by a…
Q: Which is the best reagent to accomplish this transformation? O(CH3)2C-CH2 O Ph3P-C(CH3)2 PCC 03/DMS…
A: One of the important reactions of alkene is ozonolysis. In the ozonolysis reaction, the…
Q: What is the concentration of a monoprotic weak acid if its pH is 5.50 and its ₂ = 5.7 x 10-¹⁰?
A: Given,pH of the monoprotic weak acid = 5.50Ka
Q: A student sets up the following equation to solve a problem in solution stoichiometry. (The ? stands…
A:
Q: 2. By what type of mechanism does the following aromatic substitution reaction proceed? Draw the…
A: The given reaction is an example of nucleophilic aromatic substitution reaction. The mechanism of…
Q: Calculate the pH of a solution that is 0.185M benzoic acid and 0.240M sodium benzoate, a salt whose…
A: Henderson Hasselbalch equation for acidic buffer mixture is pH = pKa + log Where pKa = - log ( Ka )…
Q: What is the major neutral organic product for the following sequence
A: Aldehyde or ketone undergoes nucleophilic addition with cyanide (CN-) to form cyanohydrin.…
Q: A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C;at this temperature it is…
A: To solve this problem, we can use the lever rule, which relates the fractions of two phases in an…
Q: Describe what happened when hydrochloric acid was added to the Cu(OH)2 precipitate. What is the…
A: When hydrochloric acid (HCl) is added to a precipitate of copper hydroxide (Cu(OH)2), a fascinating…
Q: Given the information A+ B C AG = 2D D calculate AG at 298 K for the reaction A+B 2C x10 AH = 792.8…
A:
Q: If the pH of an acidic solution at 25°C is 6.18, calculate the pOH, and the [H3O*] and [OH¯], in…
A: pH of the solution given = 6.18 pH is the negative logarithm of hydrogen ion concentration.
Q: Consider the reaction CO(g) + Cl₂(g) →COCI₂(g) Using the standard thermodynamic data in the tables…
A: The reaction CO(g) + Cl2(g) -> COCl2(g) Determine the standard Gibbs free energy change ∆G°rxn…
Q: For the reaction I2(g) +Br₂(g) 21Br(g) Ke=280 at 150 °C. Suppose that 0.520 mol IBr in a 2.00-L…
A:
Q: Complete the missing information for the following esterification: Reactant 1 + pentan-1-ol -->…
A: We have to find the missing information. Reactant 1 + pentan-1-ol --> pentyl propaanoate + water
Q: Which is the best reagent to accomplish this transformation? LOCH3 O LiAlH4 O H2/Pd-C O NaBH4 O 1.…
A: Lithium aluminum hydride (LiAlH4) is a versatile reducing reagent, that reduces the ester into…
Q: Draw a nitrogen atom, N, and an oxygen atom, O. Templates Select Draw N O More Erase
A: The atomic number of nitrogen is 7 .The atomic number of oxygen is 8 We have to draw the structure…
Q: The first five ionization energies (IE, through IE) of a Period 2 element have the following…
A: The ionisation potential shows how many valence electrons are present in an atom . Ionisation energy…
Q: What is the name of the product when pentanoic acid and methanol undergo an esterification reaction
A: Carboxylic acid reacts with alcohol to form an ester in the presence of an acid catalyst, the…
Q: escriptionSolubility (soluble, insoluble, miscible & immiscible)25 mL of water and 1 g of salt—the…
A: Solution: - A solution is a homogeneous mixture of two or more substances. A homogeneous mixture…
Q: Calculate the pH at 25°C of 239.0 mL of a buffer solution that is 0.300 M NH4Cl and 0.300 M NH3…
A: Given,Molarity of NH3 = 0.300 MMolarity of NH4Cl = 0.300 MVolume of the buffer solution = 239.0…
Q: a. b. c. Product of part b d. Br CI FeBr3 AICI3 H₂NNH₂ NaOH(aq) Na, NH3 CH3OH
A: Friedel-Crafts acylation is a classic organic reaction used to introduce an acyl group (RCO-) into…
Q: The reaction C(s)+2H2(g)⇌CH4(g)C(s)+2H2(g)⇌CH4(g) has ?p=0.263Kp=0.263 at 1000. K.1000. K.…
A: GivenKp=0.263 , Temperature=1000. K, volume=9.08 L Mass of H2=4.305 gMoles of H2=Mass of H2/Molar…
Q: Use the attached picture to help answer the question. The image shows a galvanic cell with Ag as…
A: To answer the question : The image shows a galvanic cell with Ag as one electrode and Sn as the…
Q: Which term describes a substance that acts as a proton donor and a proton acc a. Amphiprotic b.…
A: A substance that acts as a proton donor and a proton acceptor .
Q: A 21.5-liter gas cylinder contains 7.7 moles of nitrogen gas and 3.4 moles of oxygen gas. If the…
A: Volume of cylinder =21.5 LiterMoles of nitrogen gas=7.7 molesMoles of oxygen gas =3.4 molesWe need…
Q: For lead, Pb, the heat of vaporization at its normal boiling point of 1740 °C is 177.8 kJ/mol. The…
A:
Q: The pH of a solution of HCl(aq) is found to be 0.67. What is the hydronium ion concentration of this…
A: The pH is defined as the negative logarithm of hydronium ion concentration. pH = -log [H3O+] Given…
Q: A photon of radiation has a frequency of 4.64 x 108 Hz. Calculate its wavelength (in mm) and energy…
A: We have to find the wavelength whose frequency is given in the question .
Q: Nicotine is responsible for the addictive properties of tobacco. Assume Kb1 = 1.0×10-6 and Kb2 =…
A:
Q: 1. chloro 3. Inductive effect 2. -CH₂CH₂CH₂C=CH Inductive effect :0: || –CH,CH,CH,CH=CH-C—QCH3…
A: Inductive Effect: The inductive effect is a phenomenon in which the electron density is either…
Q: Highlight the atoms or groups that are in hydrophobic portions of this molecule in blue, and the…
A: Hydrophobic groups are non-polar in nature and not soluble in water.Hydrophilic groups are polar in…
Q: The decomposition of ammonia on a metal surface to form N₂ and H₂ is a zero-order reaction. At 873…
A:
Q: I want to prepare a working formulae for manufacturing a organic based body soap , for this can you…
A: Note: please keep in mind that creating and manufacturing cosmetic products requires a certain level…
Q: What are the correct reaction conditions for the following transformation? O ܐ 1. 2. H3O* 1. ܗ ' 1.…
A: Gilman reagent with the general formula R2CuLi where R may be any alkyl or aryl group.Now come on…
Q: calculate vibration energy. translational Novalicn (9) (b) also calculate He, Co₂, H₂O. BF% CHY NH3,…
A: l
Q: Exercise 3. a) Consider the oxidation of ammonia by oxygen according to the reaction Scheme: 2NH319)…
A:
Q: 4- Show how the following compounds could be synthesized starting with dimethyl malonate or ethyl…
A: Dimethylmalonate or ethyl acetoacetate and other reagents.
Q: O ELECTRONIC STRUCTURE AND CHEMICAL BONDING Identifying the electron added or removed to form an ion…
A: Electronic configuration:Electronic configuration can be define as the arrangement of electrons…
Q: The complex ion Cu(NH3)42+ is formed in a solution made0.0100 M Cu(NO3)2 and 0.400 M NH3. What are…
A:
Q: How many grams of water can be cooled from 39 °C to 19 °C by the evaporation of 44 g of water? (The…
A:
Q: The 1 five ionization energies (IE, through IES) of a Period 2 element have the following pattern:…
A: For s-block elements:Number of valence electrons = group number of elementFor p-block elements:…
Q: Name these organic compounds: compound Br CH₂-C- CH₂ - Br I C1 Br- CH3 | CH₂ - CH₂ - CH₂ - CH₂ - I F…
A:
Q: Draw the major intermediates and product of the following reaction sequence. 1.K 25 3. heat, 4.mCPBA…
A: We have to draw the major intermediates and product of the following reaction sequence.
Q: Todine-125 is radioactive and has a half life of 60.25 days, Calculate the activity of a 9.1 mg…
A:
Q: At-10.0°C, a common temperature for household freezers, what is the maximum mass of saccharin…
A:
Q: (a) Predict the significant m/z peaks in the mass spectra of the following haloalkanes. In each…
A: Mass spectrometry is useful to determine fragments, the structure of fragments and hence we can…
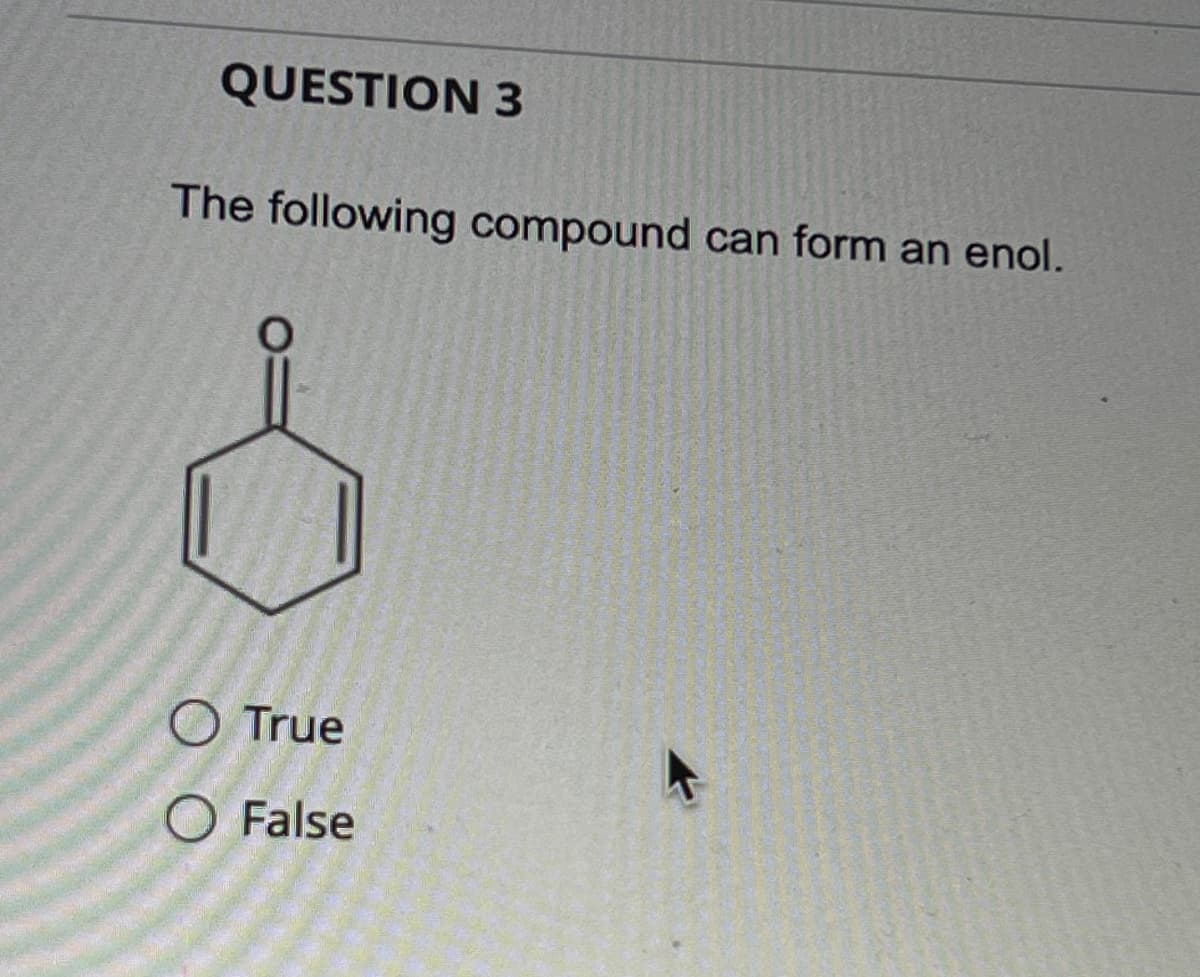
Step by step
Solved in 3 steps with 2 images

- Which is ethanal? Question 20 options: HCHO CH3CHO CH3CH2CH2CHO CH3OHPhenol is Select one: a. CH3CH2CH2OH b. C8H11OH c. CH3CH2CH2CH2CH2OH d. C6H5OHAnswer the following questions1. Give the IUPAC name of the compound in the first picture below2. Why is the reaction between these compounds not favorable (second picture ) ? State the rkind of eaction and describe why it's not favorable.
- The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate. What kind of reaction is occurring? How would you complete the synthesis?Complete the following reactions (draw the correct final products)?Write reaction of ethylmethylamine with the following reagents: a. HNO3 b. CH3COCl

