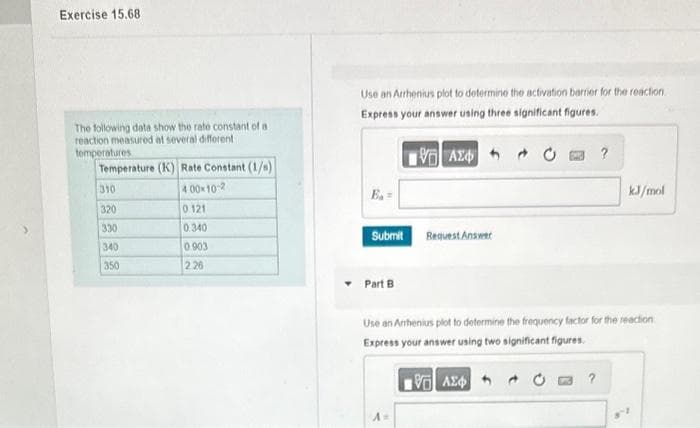
The following data show the rate constant of a reaction measured at several different temperatures Temperature (K) Rate Constant (1/s) 4.00×10-2 310 320 330 340 350 0.121 0.340 0903 2:26 Use an Arrhenius plot to determine the activation barrier for the reaction Express your answer using three significant figures. ΨΗ ΑΣΦΑ B₁- Submit Part B Request Answer A Use an Amhenius plot to determine the frequency factor for the reaction Express your answer using two significant figures. ΜΕ ΑΣΦΑ kJ/mol ? 2
The following data show the rate constant of a reaction measured at several different temperatures Temperature (K) Rate Constant (1/s) 4.00×10-2 310 320 330 340 350 0.121 0.340 0903 2:26 Use an Arrhenius plot to determine the activation barrier for the reaction Express your answer using three significant figures. ΨΗ ΑΣΦΑ B₁- Submit Part B Request Answer A Use an Amhenius plot to determine the frequency factor for the reaction Express your answer using two significant figures. ΜΕ ΑΣΦΑ kJ/mol ? 2
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter13: Rates Of Reaction
Section13.5: Temperature And Rate; Collision And Transisiton-state Theories
Problem 13.5CC: Consider the following potential-energy curves for two different reactions: a. Which reaction has a...
Related questions
Question
Transcribed Image Text:Exercise 15.68
The following data show the rate constant of a
reaction measured at several different
temperatures
Temperature (K) Rate Constant (1/s)
4.00×10-2
0.121
0.340
0.903
2.26
310
320
330
340
350
Use an Arrhenius plot to determine the activation barrier for the reaction.
Express your answer using three significant figures.
19Η ΑΣΦΑ
B₁ =
Submit
Part B
Request Answer
A=
?
Use an Amhenius plot to determine the frequency factor for the reaction
Express your answer using two significant figures.
VE ΑΣΦΑ
?
kJ/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning