The following table shows the properties of five substances. Electrical conductivity when liquid good good Melting point/°C 800 Boiling point/°C 1470 Effect of burning in air Substance solid No reaction рor good 650 1110 Burns to form a white solid Burns to form C 19 287 poor poor carbon dioxide and water Burns to form an acidic gas only No reaction D 114 444 poor рor E 1700 2200 рor poor Each substance can be used once, more than once or not at all to answer the following. Choose from A to E a substance which is: a) A metal b) A non-metallic element c) A simple molecular covalent compound d) An ionic compound e) A giant covalent substance AB
The following table shows the properties of five substances. Electrical conductivity when liquid good good Melting point/°C 800 Boiling point/°C 1470 Effect of burning in air Substance solid No reaction рor good 650 1110 Burns to form a white solid Burns to form C 19 287 poor poor carbon dioxide and water Burns to form an acidic gas only No reaction D 114 444 poor рor E 1700 2200 рor poor Each substance can be used once, more than once or not at all to answer the following. Choose from A to E a substance which is: a) A metal b) A non-metallic element c) A simple molecular covalent compound d) An ionic compound e) A giant covalent substance AB
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 134CP
Related questions
Question
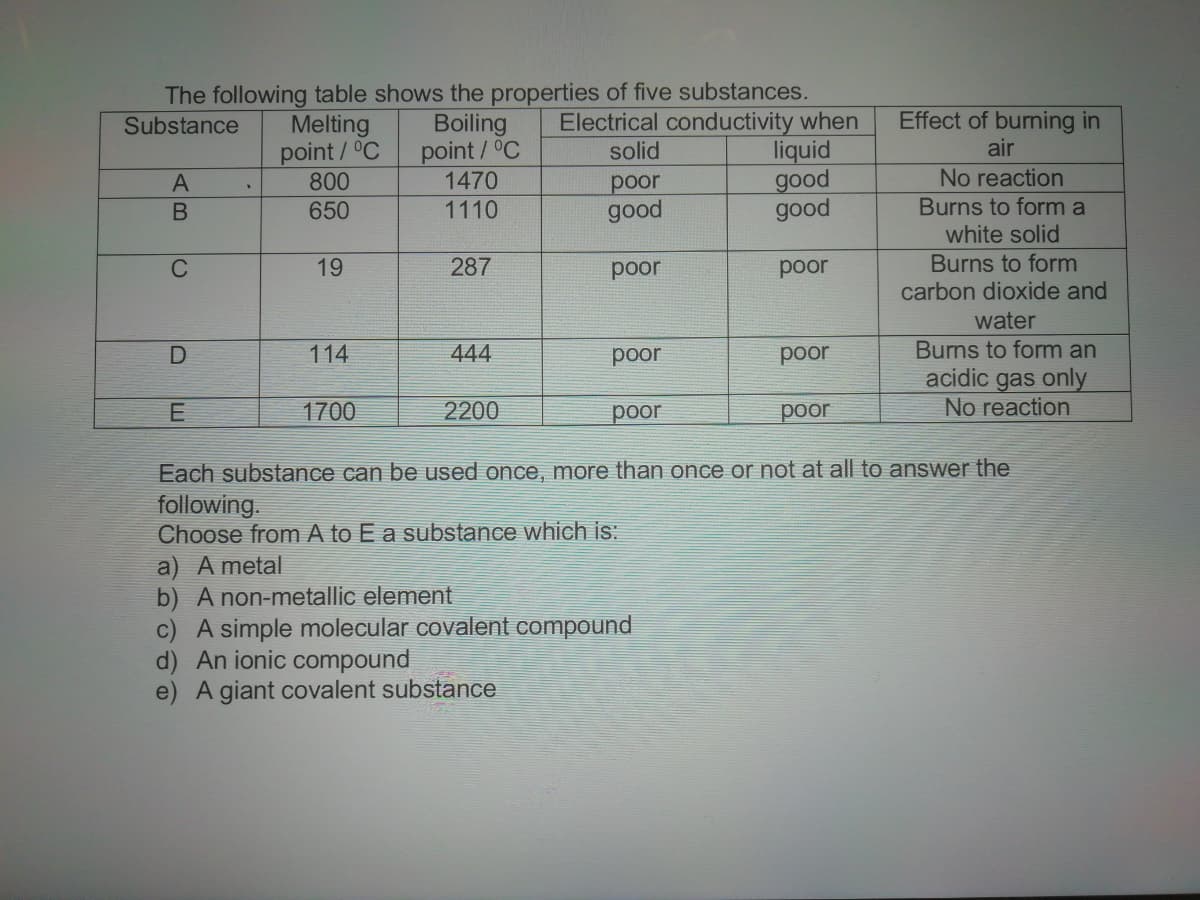
Transcribed Image Text:The following table shows the properties of five substances.
Electrical conductivity when
liquid
good
good
Melting
point/°C
800
Boiling
point/°C
1470
Effect of burning in
air
Substance
solid
No reaction
рoor
good
Burns to form a
white solid
650
1110
C
19
287
poor
poor
Burns to form
carbon dioxide and
water
Burns to form an
acidic gas only
No reaction
114
444
poor
poor
1700
2200
рoor
poor
Each substance can be used once, more than once or not at all to answer the
following.
Choose from A to E a substance which is:
a) A metal
b) A non-metallic element
c) A simple molecular covalent compound
d) An ionic compound
e) A giant covalent substance
AB
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning