Q: Give the formulas for the following covalent compounds: a) (mono)nitrogen tribromide b) dinitrogen…
A: The answer is given as follows
Q: Write the formula for iron (III) chloride trihydrate.
A: Iron symbol is Fe Chlorine symbol : Cl Fe is showing +3 oxidation state Trihydrate means , the…
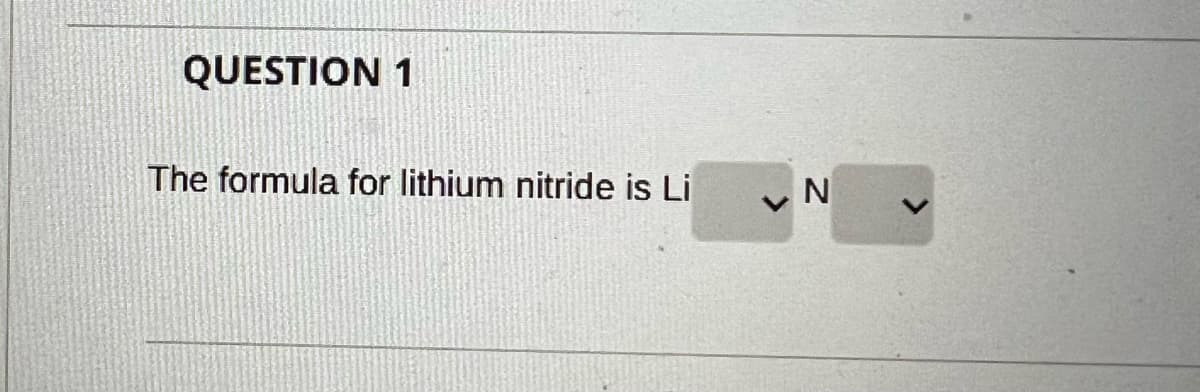
Q: Write formulas for lithium oxide, tin(II) fluoride, and lithium nitride
A: The formula of the given compounds has to be given,
Q: Write the formula for each compound. a. sodium chlorite: NaCl X b. potassium nitrite: KN02 X c.…
A: The formula for each compound can be given as follows, a. Sodium chlorite - NaClO2 b. Potassium…
Q: Write formulas for the compounds aluminum sulfide, magnesium nitride, andvanadium(III) oxide
A: Given compounds: Aluminium sulfide, Magnesium nitride, and Vanadium (III) oxide To find: The formula…
Q: Name ionic compound. PbI2
A: When a metal and a non-metal atom reacts, ionic compounds are formed. As ionic compounds consists of…
Q: Write the formula for each compound. selenium tetrafluoride: oxygen difluoride: dinitrogen monoxide:
A: Formula of the compound depends on the number of cation and number of anion
Q: Which are correct formulas for ionic compounds? Forthose that are not correct, give the correct…
A: Since you have posted multiple sub-parts, the answer for first three sub-parts are given below.…
Q: Find the formula for cadimium (I) oxide.
A:
Q: Give the systematic name for the formula or the formula for the name: (a) ammonium sulfate; (b)…
A: An ionic compound is a compound where the complete transfer of electrons takes place from one…
Q: Write the formula for osmium (IV) oxide
A: The given compound is an example of transition metal oxide. The oxidation state of a transition…
Q: Write the formula for sodium phosphate, a key ingredient in many commercial detergents.
A: Sodium has symbol Na lies in group 1 so, it has a charge of (+1). Phosphate being a polyatomic ion…
Q: Provide the formula for the compounds. chloric acid: HCIO, Incorrect lead(II) bromide: PbBr,…
A: Given that, Names of compounds given cholric acid lead(II) bromide nitrogen trifluoride We need…
Q: I. Write the formula of the compound formed by combining the ions give. ANION CATION BrO; N-3 SO,…
A:
Q: Write formulas for the compounds: (f) cobalt(II)nitrate
A: A compound is a substance which contain two or more elements of a fixed ratio in it.
Q: give the formula 1) cobalt (IV) hydroxide 2) tetraoxide trinitrogen 3) copper (V) oxide
A: The formula for these compounds can be write on the basis of their oxidation state.
Q: What is the formula for barium chloride? O Baci O BaCl2 O Ba(CIO3)2 O Ba2Cl3
A: Formula for barium chloride = ?
Q: Q12. What is the formula for manganese(III) oxide? (a) MnO (b) Mn30 (c) Mn½O3 (d) MnO3
A: Correct answer is (c) Mn2O3 . Formula of manganese(III) oxide will be Mn2O3 . Oxidation state of Mn…
Q: Write the formula for ionic compound. iron(II) phosphate
A: Chemical Formula is the short representation of the name of any chemical compound. The formula for…
Q: What is the formula for Phosphorus dioxide? O P202 O PO O KO2 O PO2
A: • We need to determine the formula for phosphorus dioxide
Q: What is the formula for calcium chlorate? O Cacl2 CaCl2 O CacIO2 OCacIO3 Ca(CIO3)2
A: Given, calcium chlorate
Q: the formula for magnesium chloride
A: Magnesium chloride is an ionic halide of magnesium. The constituent of magnesium chloride is…
Q: Give the correct formula for sodium hydrogencarbonate and indicate whether
A: sodium hydrogencarbonate is NaHCO3
Q: Name each ionic compound: (a) Na 2S; (b) AlBr 3.
A: An ionic compound is made of cation (positive ion) and anion (negative ion). Positive ions are metal…
Q: Write the formula for ionic compound. Rubidium hydrogen sulfate
A: The given ionic compound name is Rubidium hydrogen sulfate
Q: c. chlorine trifluoride CIF3 d. Iodine disulfide I,S,
A: See explanations
Q: Name the compound: K4[Ni(CN)4].
A: The compounds which have anions or neutral molecules bonded to the central metal atom by the…
Q: Write the formula for copper (II) oxide
A: The formula for copper (II) oxide is to be written.
Q: Name the ionic compound Na2S
A: Answer Name the ionic compound Na2S
Q: Write the empirical formula of at least four binary ionic compounds Ca2+,Ni4+, CI-, S2-
A: Four binary ionic compounds are :
Q: Write formula for cobalt(II) nitride Copper(III) carbonate. iron(I) sulfate.
A: NOTE: There is no such thing like iron (I) and copper (III). Hence I'm correcting it and giving the…
Q: write the formula for this compound: iridium (iii) bromide tetrahydrate
A: The formula for the compound Iridium (III) bromide tetrahydrate is IrBr3.4H2O
Q: Write the formula for ionic compound. Copper(II) chloride
A: The formula for ionic compound Copper (II) chloride is to be written.
Q: Provide the formula for the compounds. bromic acid: HBrO, lead(II) sulfide: PbS phosphorus…
A: While writing the formula of a molecule we need to first identify the each element in the compound…
Q: Consider the following reaction 2A + 3F2 → 2AF3. a. What is the formula for the reaction product if…
A: In the given reaction, 2 moles of A reacts with 3 moles of fluorine to form 2 moles of AF3.
Q: What is the formula of C+4 and O-2 N+3 and Cl-1
A: We are given two ions that can show electrostatic interactions and a iinic bond is formed between…
Q: Name the compound Fe(NO3)2
A: Introduction : When two or more atoms of different elements combine together in a definite ratio…
Q: ionic compound cation anion NaCl Na CI CuS O4 NİCI, c:(NO.), NH,I
A: The metal part is the cationic part and the remaining part is anionic part and NH4+ is also an…
Q: Name ionic compound. PbI4
A: Pb is the symbol for Lead and I is the symbol for Iodine. The oxidation state of Lead in this given…
Q: Complete the table below and name the compound formed. so? PO lons co, 4 4 ,+3 Al NH +1 4 +2 Са ,+1…
A: Ions CO32- Cl- SO42- PO43- Al3+ Al2(CO3)3 aluminium carbonate AlCl3 Aluminium trichloride…
Q: Write the formula for calcium nitride
A: The chemical formula of any compound consists of the electropositive part at the left side and the…
Q: Write the formula for sodium disulfomercurate (II). C Naz[HgS] C Na[HgS,]+2 O Na[HgS] C Na[HgS2]…
A: In this question, we will see the correct formula for sodium disulfomercurate (II) You can see the…
Q: Write the formula for strontium sulfide
A: Given compound is strontium sulfide. Strontium is an example for a transition metal which means that…
Q: The compound MgCl2 is named dimagnesium chloride. magnesium chlorine. magnesium (II) chloride.…
A: MgCl2 is an inorganic and ionic compound . It consists of ions of magnesium and ions of chlorine.
Q: 2. The formula for Ferric chloride is......... a) FeCl b) FeCl3 c) CuCl3 d) FeCl2
A:
Q: What is the formula for Potassium Hydroxide? А. КОН о в. К(ОН)2 O C. K2HO O D. KO
A: Formula of a compound is written by the combination of basic radical first then acidic radical
Q: Provide the name for CoN. Cobalt nitrate Cobalt nitride Carbon oxygen nitride Cobalt (III) nitride
A: The name of CoN is Cobalt nitrate.
Q: The correct formula for chromium (III) perchlorate is: Cl3CrO12 OTrue False
A: Since the formula for perchlorate is ClO4- And since in the compound name its given that Cr i.e…
Q: name the ionic compound PbCl4
A: The name of an ionic compound is written as follows: First, write the name of the cation is…
Q: Write the formula for tin(IV) oxide.
A: A covalent compound is a compound that is formed by the complete sharing of electrons between the…
Step by step
Solved in 2 steps

- Question 11 What formula/name combination is correct? Group of answer choices Chromium(III) sulfide is Cr2S3 Zinc(II) oxide is Zn2O Sulfur(VI) oxide is SO3 Iron trichloride is FeCl3 None of these answers is correctChoose the combination of formula/name that is incorrect: Group of answer choices CS2 is carbon disulfide Mg(NO3)2 is magnesium nitrate H2SO4(aq) is sulfuric acid FeCl3 is iron chloride HC2H3O2 is acetic acidFind the formula for cadimium (I) oxide.

