The graph here shows the pH curve for the titration of 25.00 mL of a 0.100-M monoprotic acid solution, HA(aq), with a 0.1 M solution of a monoprotic base. 14 Hd 12 8 2. 10 15 20 25 30 35 40 45 50 Volume of 0.100-M base added (mL) (1) The pH curve represents the titration of a acid with a O base. (2) Choose a suitable indicator for the endpoint of the titration from the following pulldown lis v to the table of indicators in the Approach. Refer Bromocresol green Methyl red Bromothymol blue Submit Show Approach Show Tutor Steps Cresol red Thymol blue Phenolphthalein
The graph here shows the pH curve for the titration of 25.00 mL of a 0.100-M monoprotic acid solution, HA(aq), with a 0.1 M solution of a monoprotic base. 14 Hd 12 8 2. 10 15 20 25 30 35 40 45 50 Volume of 0.100-M base added (mL) (1) The pH curve represents the titration of a acid with a O base. (2) Choose a suitable indicator for the endpoint of the titration from the following pulldown lis v to the table of indicators in the Approach. Refer Bromocresol green Methyl red Bromothymol blue Submit Show Approach Show Tutor Steps Cresol red Thymol blue Phenolphthalein
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 74QAP: Fifty cm3 of 1.000 M nitrous acid is titrated with 0.850 M NaOH. What is the pH of the solution (a)...
Related questions
Question
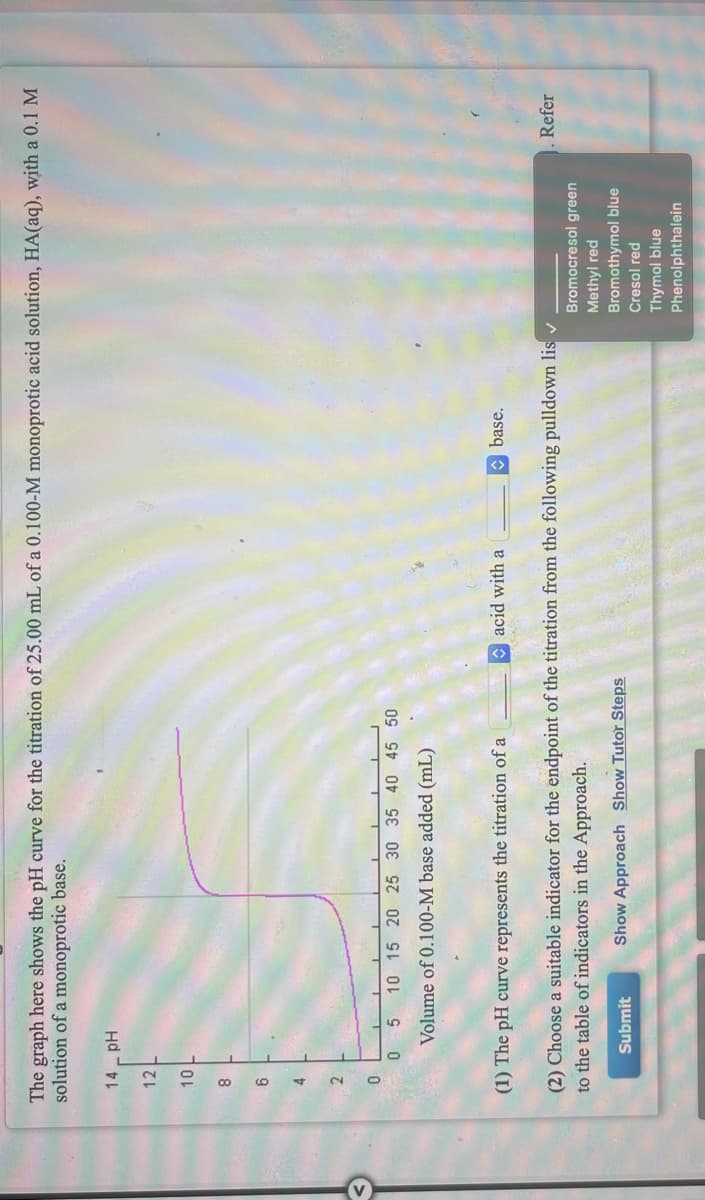
Transcribed Image Text:The graph here shows the pH curve for the titration of 25.00 mL of a 0.100-M monoprotic acid solution, HA(aq), with a 0.1 M
solution of a monoprotic base.
Hd
12
8
9
2
10 15 20 25 30 35 40 45 50
Volume of 0.100-M base added (mL)
(1) The pH curve represents the titration of a
acid with a
O base.
(2) Choose a suitable indicator for the endpoint of the titration from the following pulldown lis v
to the table of indicators in the Approach.
Refer
Bromocresol green
Methyi red
Bromothymol blue
Submit
Show Approach Show Tutor Steps
Cresol red
Thymol blue
Phenolphthalein
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning