Q: The thermal energy of 0.900 mol of a substance is increased by 0.900 J.
A:
Q: How much heat is absorbed by a 2kg iron skillet over a camp fire as it goes from 25*C to 150*C,…
A: The specific heat of iron is 450 J/kg°C. Write the equation for the heat absorbed. Q=mCTf-T1 Here,…
Q: A change in temperature of 50°C is equal to a change of?
A: Change of temperature in degree Celsius and kelvin is same.For example
Q: How much energy is required to heat 36 kg of steel (c=0.490 kJ/(kg°C)) from 81.9°C to 1230°C for…
A: Write the equation for the specific heat. Q=mc∆T Here, The specific heat is "Q" The change in the…
Q: Thirty-two joules of heat is added to a 255gram iron bar How much will the iron bar’s temperature…
A: Amount of Heart added = 32 J Mass of iron bar = 255 g Specific heat of iron bar, C=0.46 J/g o C
Q: A silver bar has a mass of 0.525 kg. When 1230 joules of thermal energy is added to the bar, its…
A: The specific heat capacity of silver is c = 234.286 J/kg* 0C
Q: After the 1 kg block of ice melts, the amount of heat required to raise its temperature to 10°C is…
A: Given, Mass of melted ice(water), m= 1kg, increase in temperature, ∆T=100 C=283.15 K.
Q: The temperature of 100 g of water is to be raised from 10 c to 90 c. The energy needed to do this is…
A: Given data: Mass m =100g Temperature T1=10°C Temperature T2=90°C
Q: Two substances are warmed from 0°C. Their temperatures are plotted as a function of the amount of…
A: The specific heat of a substance is the amount of energy required to raise the temperature of one…
Q: How much energy is required to heat 36 kg of steel from 81.9°C to 1230°C for forge welding?
A: Given quantities: mass of the steel (m) = 36 kg initial temperature (T0) = 81.9 degree Celsius final…
Q: How much heat is removed when 100 g of steam at 150◦C is cooled and frozen into 100 g of ice at 0◦C.…
A:
Q: The temperature of a 50 g sample of aluminum is raised from 20oC to 90oC when 770 cal of heat is…
A: Given,
Q: uired to raise the temperature of 4.1 kg of ice at -31.°C to water a
A: Given:- The mass of the ice m = 4.1 kg The change in temperature T1 = -31 oC = -31+273 = 242 K T2…
Q: A 69g block of metal has a specific heat of 1.29J. What will be the temperature of this metal to one…
A: Given :- mass of block m = 69 g = 0.069 kg specific heat C = 1.29 J heat energy Q = 114 J…
Q: How much energy is required to heat 31.3 kg of steel (c=0.490 kJ/(kg°C)) from 45.6°C to 1230°C for…
A: Given: Mass, m=31.3 kg Ti = 45.6°C Tf=1230°C∆T=1184.4°Cspecific heat capacity of liquid…
Q: The amount of heat required to raise the temperature of 2 kg of iron (sp. Heat = 0.115 kcal/kg-C)…
A:
Q: If 5 kg of water freezes at 0ᵒ C, how many kilocalories of heat are liberated?
A: Amount of heat release when water freezes is called latent heat of fusion. Latent heat of fusion(L)=…
Q: An automobile cooling system holds 16 L of water. The specific heat of water is 4186 J/kg⋅C. How…
A:
Q: A bottle containing 6 kg of water at a temperature of 15°C is placed in a refrigerator where the…
A:
Q: A hot 1 kg chunk of copper is allowed to cool from 700°C to 100°C. If the specific heat of copper is…
A: Given data The mass of the chunk of copper is: m = 1 kg = 1000 g The initial temperature of the…
Q: energy has been added in joules
A:
Q: The heat of 50MJ in joule is ?
A: 50 MJ = 50 x 106 J or 50 MJ = 50000000 J or 50 MJ = 50,000,000 J
Q: How much will the temperature change if 5000J of heat is added to 1.0kg of sand?
A: Given data: Heat added (Q) = 5000 J Mass (m) = 1.0 kg Required: Change in temperature (ΔT)
Q: The thermal energy of a container of helium gas is halved. What happens to the temperature, in…
A: The expression for average kinetic energy for a gas is KEavg=32kBT Average kinetic energy of a gas…
Q: 567J of heat is absorbed by a 2 kg iron skillet over a camp fire as it goes from 25°C to 150°C,…
A: Introduction: Specific heat capacity: The specific heat capacity of a substance is defined as the…
Q: A change in temperature of 50 ^ 0 C is equal to a change of
A:
Q: How much heat must be removed when 50 grams of steam at 100°C Is condensed to form liquid water at…
A:
Q: The amount of heat needed to raise the temperature of 1 gram of water by 1°C is
A: the amount of heat required at a pressure of 1 standard atmosphere to raise the temperature of 1…
Q: What is the amount of energy required to raise the temperature of a 2.5 kg block of ice at -25°C to…
A:
Q: how much heat is needed to raise the temperature of 2 kg of water 5k
A:
Q: 0.025 kg of mercury is heated from 25°C to 155°C, and absorbs 455 joules of heat in the process.…
A:
Q: How much heat is required to convert ice with a mass of 12.0g at an initial temperature of -10.0°C…
A: For heating ice from -10oC to 0oC : The heat required is Q1=mc∆TQ1=12(2.1)(0-(-10))Q1=252 J
Q: A person consumes about 2300 Cal a day. 1 Cal = 4186 J and 1 kWh= 860 Cal.If a power company charges…
A: person consumes = 2300 cal in one day 1 kwh = 860 cal cost of 1 kwh energy = 12 ¢ means cost of 860…
Q: 50 g of copper is heated to increase its temperature by 100C. If the same amount of heat is applied…
A:
Q: Calculate the amount of heat (in kJ) needed to raise the temperature of 50.0g of water 20.0°C to…
A:
Q: How much heat joules is needed of 5.5 L of water to raise the temperature of of water from 0°C to…
A: Given- Volume of water , V = 5.5 L Initial temperature of water, Ti = 0o C Final temperature of…
Q: If 25 kJ is necessary to raise the temperature of a block from 25C to 30C, how much heat is…
A:
Q: The water equivalent of 5 kg of a substance is 1 kg. By how much will its temperature change if 7550…
A:
Q: The temperature of 100g of water is raised from 10 celsuis to 90 celsuis. The energy needed to be…
A:
Q: 10 kg of copper wire is heated from room temperature (22 C) to 45 C. What was the change in Thermal…
A: Given, Mass of rod m = 10 Kg T0 = 22C∘ Tf = 45C∘
Q: How much heat (in J) is needed to raise the temperature
A: Given that:- Mass=9kg T1=380 C, T2=8300 C /\T=T2-T1=7920 C C= Specific heat capacity of silver=230…
Q: What is the specific heat of a 5.1-kg substance if 135 J of heat is needed to raise the temperature…
A: Given : Mass m=5.1 kg Heat Q=135 J Temperature T1=283.15 K Temperature T2=303.15 K
Q: If 25 kJ is necessary to raise the temperature of a block from 25ºC to 30ºC , how much heat is…
A: The expression for the required necessary heat,
Q: One gram of ice at 0°C is placed on a container containing 2,000,000 cubic meters of water at 0°C.…
A: Given data: ice's mass m = 1 g Initial temperature Ti = 0 oC final temperature Tf = 0 oC volume of…
Q: You take an ice-cream out of the freezer, kept at −18 ∘C. Outside it is 32 ∘C. After one minute, the…
A: Explanation We will solve the problem using Newton's law of cooling. From this law we know rate of…
Q: A metal is heated until its temperature rises from 8℃ to 45℃. Calculate the heat capacity if the…
A:
Q: The amount of heat needed to raise 1g of water by 1c is?
A: Specific heat of substance is the amount of heat required to raise the temperature of 1 g mass by…
Q: If 2059 J of heat is gained by 57 g of oil that has specific heat capacity of (2.4 J/(g °C),…
A: Heat gained = 2059 Joules Mass of oil = 57 g Specific heat capacity of oil = 2.4 J/(g-°C)
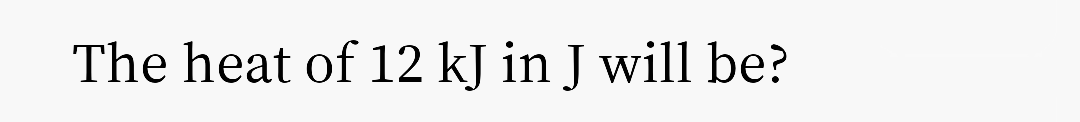
Step by step
Solved in 2 steps with 2 images

- How much heat is absorbed by a 2kg iron skillet over a camp fire as it goes from 25*C to 150*C, irons specific heat is...?How much heat (in J) is needed to raise the temperature of 7 kg of iron from 22 degrees Celsius to 810 degrees Celsius?A change in temperature of 50 ^ 0 C is equal to a change of
- What is the specific heat of a substance if 120 kJ of heat is needed to raise 4.1 kg of the substance from 18 degrees Celsius to 37.2 degrees Celsius?How much heat joules is needed of 5.5 L of water to raise the temperature of of water from 0°C to 81.0°C?If you have 500 g of water at 25 degrees celcius and wish to heat it to 74 degrees Celsius, how much heat is required and what is the specific heat of water?
- How much will the temperature change if 5000J of heat is added to 1.0kg of sand?Two substances are warmed from 0°C. Their temperatures are plotted as a function of the amount of heat added. Which substance has a larger specific heat?What is the amount of energy required to raise the temperature of a 2.5 kg block of ice at -25°C to steam at 100°C My answer was 8,275,000J



