Q: CH3 + NAOH CH CH CH CH A) II B)I C) II D) IV Which carbon atoms are most susceptible to nucleophilic…
A: Given, reaction is the acid-base reaction. Electron withdrawing groups like carbonyl group (CO)…
Q: What functional group would be synthesized through the following reaction? CH,CH,CH, + 2CH,OH a.…
A: Given is reaction of ketone with alcohol.
Q: Which of the following exists in mainly the enol tautomeric form? H3C H H3C CH 3 I H3C "I CH3 EtO…
A: Generally carbonyl compunds exits in keto form because keto form is more stable than enol form. But…
Q: What is the formula of the hemiacetal of methanol and propanal? A CH3CH2CH(OCH3)OH CH3CH(OC2H5)OH…
A: given methanol - CH3OH propanal - CH3CH2CHO
Q: Which of the following is the major organic product in the reaction sequence? CH;CH,M9CI diethyi…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: 1. Rank each set of three alcohols in terms of increasing acidity, "1" being least acidic, and "3"…
A: - Inductive effect increases the acidic strength of the compound. More is the number of…
Q: What is required to complete the following reaction? O NaOH/H₂0 O 1) PPH3 CH2 2)NaBH4 O 1) CH3MgBr.,…
A:
Q: Draw the structural formula of the enol formed in each alkyne hydration reaction; then draw the…
A:
Q: You identified aldehydes or ketones by converting them into hydrazones. Which of the following…
A:
Q: Show how to convert propene to each of these compounds, using any inorganic reagents as necessary.…
A: Since you have asked question with multiple subparts, we will solve the first 3 subparts for you, If…
Q: Propyl alcohol will be oxidized to propanaldehyde using O I. KMNO4 II. NaBH4 III. LIAIH4 IV. PCC
A: Given: Oxidation of propyl alcohol to propanaldehyde.
Q: Which of the following carboxylic acids has the highest solubility in water? A) CH₃CH₂C(=O)OH B)…
A: Solubility of any substance is the property to dissolve completely in the solvent. The concept of…
Q: Identify the Type of Product 1-propene + H2O—> 1-propanol a. zaitsev b. markovnikov c.…
A: Alkenes can readily react with electrophiles. Markonikov rule states that the positive part is added…
Q: Which of the following statements is true about the organic product of this reaction if the reaction…
A: Given reaction is , addition reaction of grignard reagent on ketone.
Q: HO HO %3D
A: Dehydration reaction: A chemical reaction whereby a water molecule is lost.
Q: 2) Name the following alcohols and phenols using the IUPAC and common nomenclature (If applies). OH…
A: A question based on nomenclature, which is to be accomplished.
Q: Draw the structural formula of the enol formed in each alkyne hydration reaction; then draw the…
A:
Q: Which of the reagents listed below can convert a primary alcohol to an aldehyde? O AGNO3 O H2Cr04…
A: The oxidation reaction of alcohols are reagent dependent which means that strong reagent leads to…
Q: 5. Which of the following would not yield the alcohol shown below? он а. 1. „MgBr 2. H;O* b. 1.…
A: An organo Lithium Reagent is R-Li and a Grignard Regent is R-MgX, R is an alkyl group and X is a…
Q: What is the major organic product of the following reaction? CH3-CH-CH,CH3 CH;CH,ÖH room temperature…
A: Ethanol(CH3CH2OH) is considered as a weak nucleophile which follow SN1 mechanism
Q: 9. Which of the following would show the most similar chemical behavior to 1-butanol, which is shown…
A:
Q: H H. H.
A: Phosphorus tribromide (PBr3) and phosphorus trichloride(PCl3) are reagents that are used to…
Q: Which of the following is the major organic product in the reaction sequence? 1. CH;CH,MGCI H3C-CECH…
A: The grignard reagent reacts with terminal alkyne to form an alkane and another Grignard reagent.
Q: Pick the best IUPAC name for the following molecule: H3C-O CH3 CH3 H3C– O 2,4,5-trimethylheptanal…
A: The correct iupac name of the compound is given below
Q: Predict the major products formed when benzene reacts (just once) with the following reagents.…
A: This is a friedel craft reaction in which benzene reacts with alcohol in the presence of lewis acid…
Q: Which of the following aldehydes has the highest solubility in water?
A: Solubility of aldehyde is due the carbonyl group . As we extend the number of carbon it's solubility…
Q: Give the structures of the products you would expect when each alcohol reacts with (1) HCl, ZnCl2;…
A:
Q: Which of the following will produce 2-methyl-2-butanol? 1) xs CH3M9B I. 2) H20 1) CH3MgBr I. 2) H20…
A: The products of the given reactions are as follows:
Q: NH2 aniline trans- H. cinnamaldehyde OH 3-methylcyclohexanol CH3 diphenylmethane ethyl acetate H3C…
A: In the given question we have to identify the functional group in the following compound. 1-…
Q: 1. Predict the products, if any, for the reaction of benzene with the following reagents: a.…
A:
Q: When trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product.…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 5-ethyl-2-octanone 4-ethylheptanoyl chloride O CH3LI O CH3MgBr O (CH3)2CuLi O 1. Mg/ether 2. CH3Br O…
A: Here we have to convert acid chloride group to Ketone group. Gillman reagent ( R2CuLi ) is best…
Q: (a) rate at which the following alcohols undergo dehydration to form alkenes in sulfuric acid OH OH…
A: Given : structure of molecules Rate is directly proportional to the stability of formed carbocation.…
Q: 1. STRUCTURE-DESCRIPTION Draw the structure of a compound that fits the given description. 1. the…
A: Alcohol are the -OH group containing compounds. It's solubility in water is depends on the alkyl…
Q: How are the following conversions carried out?(i) Propene → Propan-2-ol(ii) Benzyl chloride → Benzyl…
A: (i) Propene → Propan-2-ol
Q: Which of the following compounds (i through v) should not be classified as an acetal ? H CH30- OCH3…
A:
Q: Dehydration of alcohols in the presence of strong acid follows the O A. Markovnikov's rule O B.…
A: Dehydration of alcohols in the presence of a strong acid such as sulfuric acid involves removal of a…
Q: 6. Complete the following halogenation reactions for alcohols. Draw the structure of the product.…
A:
Q: 2-methylhexanoic acid can be synthesized by oxidation of which of the following compounds? A)…
A: Oxidation reactions are those reactions in which the addition of oxygen takes place using an…
Q: Q. Which of the following sets gives sec-butyl alcohol with water in an acid medium? (i) 1-butene…
A: When 1-butene is treated with water, in acidic medium then sec- butyl alcohol (2-butanol) is formed…
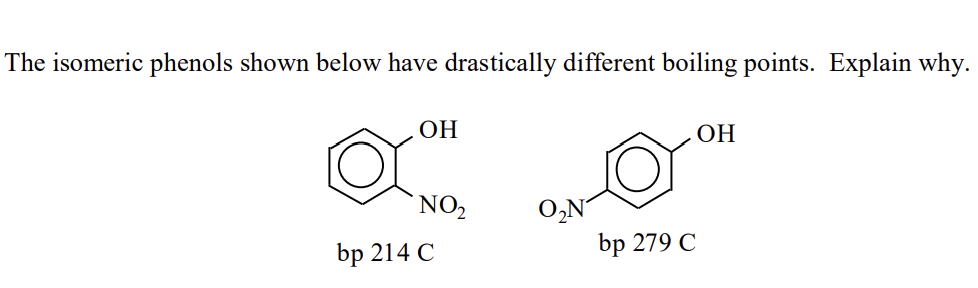
Q: Of the following type 2 carbonyl compounds below, which one had the highest boiling point? OH `NH2…
A: Boiling point is the temperature at which a substance converts to vapors from liquid state. The…
Q: Markovnikov's rule is needed to predict the product in the reaction between HCl and which compound…
A: Addition of a protic acid (HX) to the assymetric alkene or alkyne where X group is attached to more…
Q: 5. Which one of the following alkenes will produce a secondary alcohol as a major product when…
A: The organic compounds in which two carbon atoms are bonded to each other by a double bond are called…
Q: Which one of the following compounds give racemic mixture when treated with NaOH (aq)? A…
A: To obtain racemic mixture we know that the reaction should proceed through SN1 reaction mechanism.…
Q: Which of the following compounds can react with C2H5MgBr to give 3-pentanol? a.acetone…
A: C2H5MgBr is named ethyl magnesium bromide, it is an example of a Grignard reagent. It is a special…
Q: 7) Which structure below represents a ketone? A) CH3CH2 0– CH,CH3 B) II C- Он C) CH3CH2C H D) -ċ-CH3…
A:
Q: Which of the following compounds does not form an alcohol when it reacts with excess Grignard…
A: The given compounds of A, B and C are given below - The given compound A is an amide. This compound…
Q: Which of the following compounds would be most soluble in water? c. CH-CH-CH-CH-OH a. CH;-CH-CH C-H…
A: Solubility of a compound follows the rule of like dissolves like. This means that polar solute…
Q: Which of the following alcohols cannot be obtained by adding water to alkenes? 1111E Methanol II-1…
A: Addition of water to alkene goes via carbocation intermediate, only a stable (30, 20, conjugated)…
Q: HO, 1,2-dihydroxybenzene 7) 8) CH CH, HO CH 3,44-triphenyl-2-butanol 10)
A:
Step by step
Solved in 2 steps

- Give reasons for the following :(i) Ethyl iodide undergoes SN2 reaction faster than ethyl bromide.(ii) (±) 2-Butanol is optically inactive.(iii) C—X bond length in halobenzene is smaller than C—X bond length in CH3—X.How will you convert the following compounds to benzene?(i) Acetylene (ii) Benzoic acid(iii) Cyclohexane (iv) Benzene diazonium chloride.When 1-butanol is treated with conc. H2SO4 and heat .A product 'A' is formed from the reaction. Treatment of 'A' with HCl/H2O gives 'B'. Treatment of 'A' with cold KMnO4/OH- gives 'C' Treatment of 'A' with hot KMnO4 /OH- gives 'D' followed by acidification of the mixture to give 'E' 1. identify the compounds represented by A,B,C,D and E.
- What product is obtained from the reaction of excess benzene with each of the following reagents? a. isobutyl chloride + AlCl3 b. 1-chloro-2,2-dimethylpropane + AlCl3 c. dichloromethane + AlCl3What happens if toluene is treated with: a) Br2/UV, b) Cl2/AlCl3, c) HNO3conc./H2SO4conc., d) (CH3)2CHBr/ZnBr2, e) KMnO4/H+, t°C. Name all the products.Which of the following reactions will synthesize phenol from benzene? 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) warm H2SO4 and H2O 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) CuCN; 5) dilute acid and heat 1) Acetyl chloride & AlCl3; 2) bleach 1) Ph-N2+ + KI; 2) BrMgCH=CH2 in ether, followed by H3O+; 3) warm, conc'd KMnO4 1) Cl-CH(CH3)-CH2CH2CH3 + FeBr3; 2) hot, conc'd KMnO4
- Write reactions of propan-1-ol with the following reagents: a. Na b. PCl5Give reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.a. Compound X is benzene, Y is acetic anhydride acid. Complete the following scheme and determine Z! b. Determine which reagents except acetic acid anhydrides can replace Y!
- 2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (i) Draw the structural formula of compounds W, X and YChoose the appropriate reagent for the following reaction. A) TsCl/pyridine B) SOCl2/pyridine C) ZnCl2 D) PBr3 E) HIPredict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. (a) PBr3 (b) SOCl2 (c) Lucas reagent

