Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter4: Recrystallization
Section: Chapter Questions
Problem 1Q
Related questions
Question
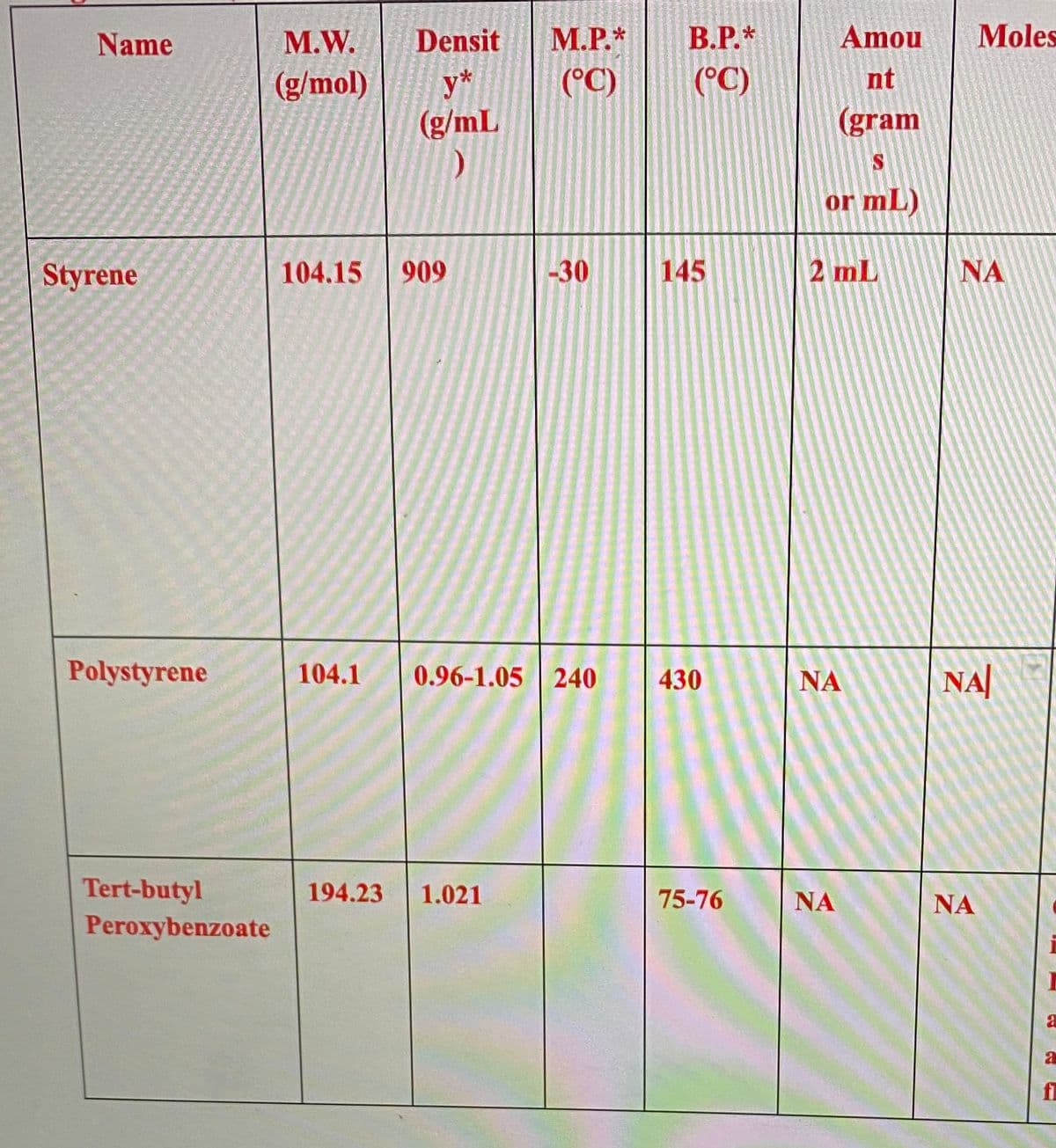
Transcribed Image Text:Name
Styrene
Polystyrene
Tert-butyl
Peroxybenzoate
M.W. Densit M.P.* B.P.*
(g/mol)
(°C) (°C)
y*
(g/mL
104.15 909
-30
104.1 0.96-1.05 240
194.23 1.021
145
430
75-76
Amou
nt
(gram
S
or mL)
2 mL
ΝΑ
ΝΑ
Moles
ΝΑ
NA
ΝΑ
a
fl

Transcribed Image Text:The limiting reagent is
Calculating theoretical yield:
Molar mass of styrene
Moles of styrene = |
The theoretical yield is
Expert Solution
Step 1
The reaction is the polymerization of styrene using the initiator Tert-butyl Peroxybenzoate.
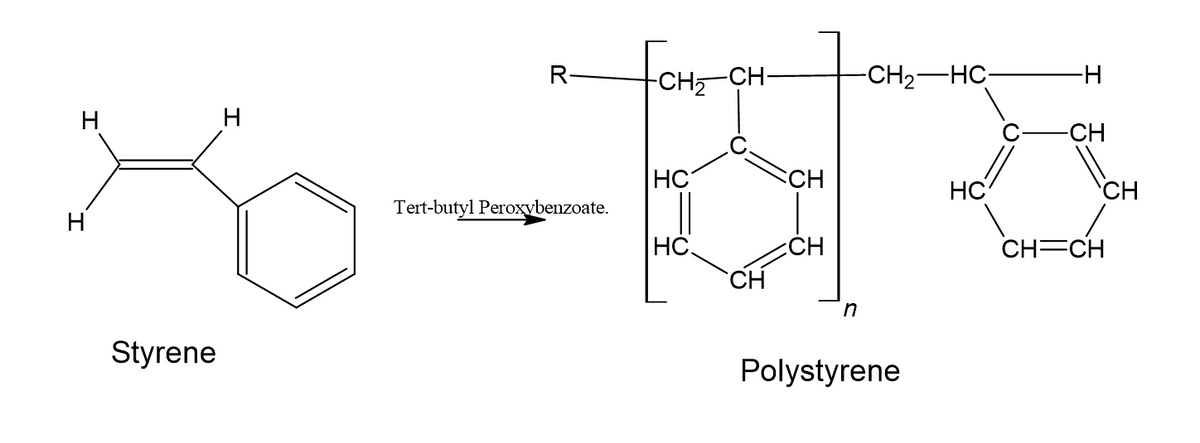
In this reaction the yield of polystyrene is dependent on the amount of styrene.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole