The mass difference between two isotopes is sometimes just a neutron mass. The spectrometer should separate them very well. For such an isotope combination, the difference in radius should be around 1 cm. That is r, -r = 1 cm. In order to achieve this, choose a magnetic field with a magnitude in Tesla (maximum magnetic field you can obtain from a conventional magnet is around 2.5 T so be far away from this value) and choose the direction also. Then determine the velocity of isotope you need. Last calculate radius r of a Pbo4 smallest isotope.
The mass difference between two isotopes is sometimes just a neutron mass. The spectrometer should separate them very well. For such an isotope combination, the difference in radius should be around 1 cm. That is r, -r = 1 cm. In order to achieve this, choose a magnetic field with a magnitude in Tesla (maximum magnetic field you can obtain from a conventional magnet is around 2.5 T so be far away from this value) and choose the direction also. Then determine the velocity of isotope you need. Last calculate radius r of a Pbo4 smallest isotope.
Related questions
Question

Transcribed Image Text:The mass difference between two isotopes is sometimes just a neutron
mass. The spectrometer should separate them very well. For such an
isotope combination, the difference in radius should be around 1 cm. That
is r, -r = 1 cm. In order to achieve this, choose a magnetic field with a
magnitude in Tesla (maximum magnetic field you can obtain from a
conventional magnet is around 2.5 T so be far away from this value) and
choose the direction also. Then determine the velocity of isotope you need.
Last calculate radius r of a Pbo4 smallest isotope.

Expert Solution
Step 1
Radius of circular path taken by a charged particle when placed in a magnetic field
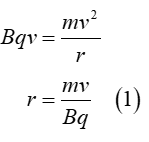
Step 2
Difference in radii of two isotopic ions
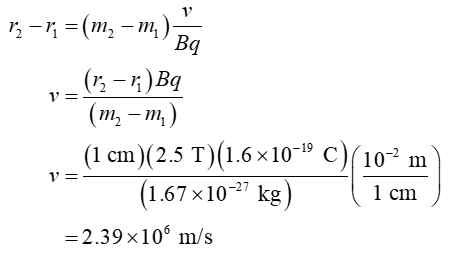
Step by step
Solved in 3 steps with 3 images
