The molecular formula of aspartame, the artificial sweetener marketed as NutraSweet, is C14H18N2O5. How many moles of aspartame are present in 4.50 mg of aspartame? Express your answer using three significant figures. n= VO ΑΣΦ MERINE SELEC ? mol
The molecular formula of aspartame, the artificial sweetener marketed as NutraSweet, is C14H18N2O5. How many moles of aspartame are present in 4.50 mg of aspartame? Express your answer using three significant figures. n= VO ΑΣΦ MERINE SELEC ? mol
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 54P: (a) Using data from the previous problem, find the mass of nitrogen, oxygen, and argon in 1 mol of...
Related questions
Question
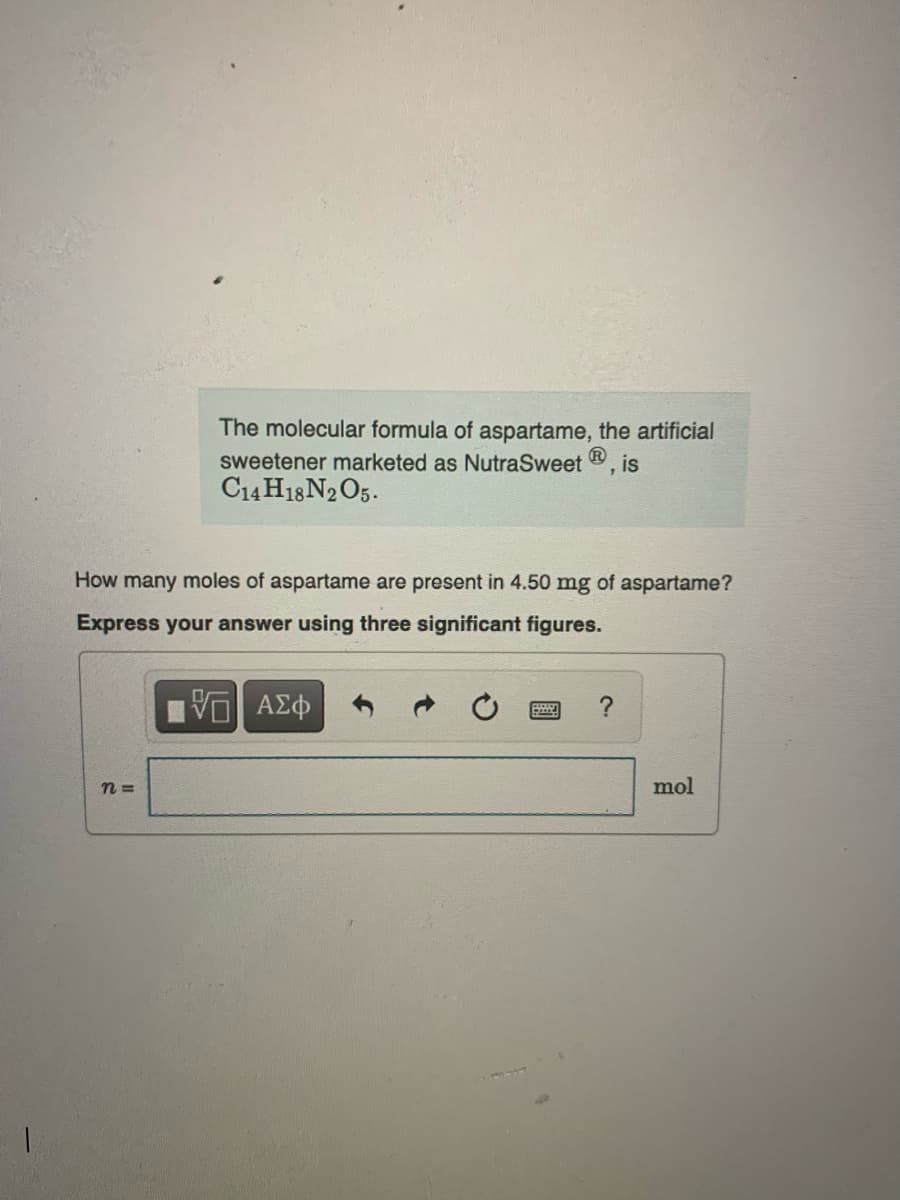
Transcribed Image Text:The molecular formula of aspartame, the artificial
sweetener marketed as NutraSweet Ⓡ, is
C14H18 N2O5.
How many moles of aspartame are present in 4.50 mg of aspartame?
Express your answer using three significant figures.
| ΑΣΦ
n=
?
mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning