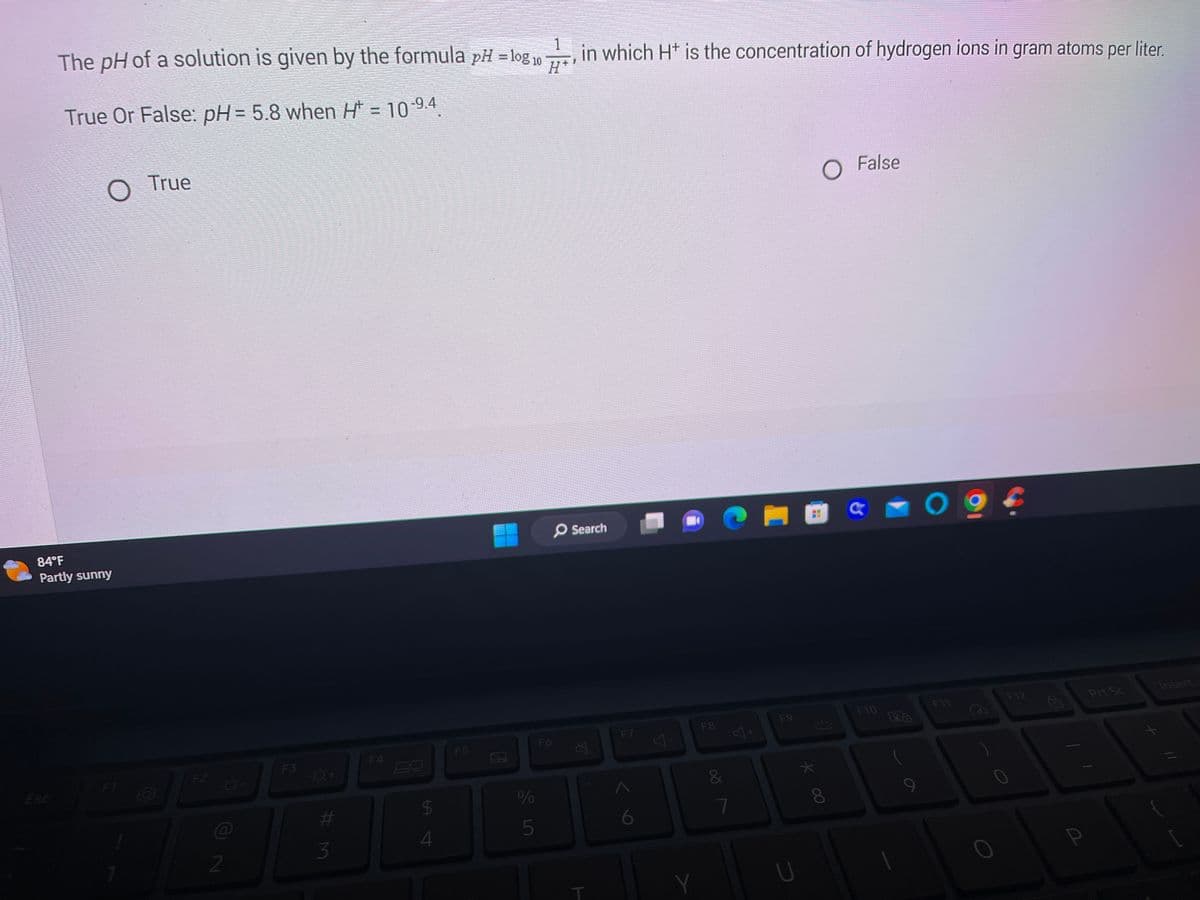
The pH of a solution is given by the formula pH = log 10- in which H+ is the concentration of hydrogen ions in gram atoms per liter. H* True Or False: pH= 5.8 when H = 10-9.4 True False
The pH of a solution is given by the formula pH = log 10- in which H+ is the concentration of hydrogen ions in gram atoms per liter. H* True Or False: pH= 5.8 when H = 10-9.4 True False
Chapter6: Exponential And Logarithmic Functions
Section6.6: Exponential And Logarithmic Equations
Problem 79SE: Recall the formula for continually compoundinginterest, y=Aekt. Use the definition of a...
Related questions
Question
Transcribed Image Text:1
The pH of a solution is given by the formula pH = log 10
in which H+ is the concentration of hydrogen ions in gram atoms per liter.
H*
True Or False: pH = 5.8 when H* = 10-9.4.
Esc
O True
84°F
Partly sunny
F1
F2
2
F3
-O+
#M
3
F4
$
4
F5
%
5
F6
Search
X
T
F7
6
A
Y
F8
&
7
S+
F9
★
* 00
O False
€1
F10
O
F11
0
F12
fal
Prt Sc
Insert
//
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 7 images

Recommended textbooks for you


College Algebra
Algebra
ISBN:
9781305115545
Author:
James Stewart, Lothar Redlin, Saleem Watson
Publisher:
Cengage Learning

College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning


College Algebra
Algebra
ISBN:
9781305115545
Author:
James Stewart, Lothar Redlin, Saleem Watson
Publisher:
Cengage Learning

College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning

Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage

Functions and Change: A Modeling Approach to Coll…
Algebra
ISBN:
9781337111348
Author:
Bruce Crauder, Benny Evans, Alan Noell
Publisher:
Cengage Learning
