The pressure, volume, and temperature of a mole of an ideal gas are related by the equation PV = 8.317, where P is measured in kilopascals, V in liters, and T in kelvins. Use differentials to find the approximate change in the pressure if the volume increases from 10 L to 10.6 L and the temperature decreases from 340 K to 330 K. (Note
The pressure, volume, and temperature of a mole of an ideal gas are related by the equation PV = 8.317, where P is measured in kilopascals, V in liters, and T in kelvins. Use differentials to find the approximate change in the pressure if the volume increases from 10 L to 10.6 L and the temperature decreases from 340 K to 330 K. (Note
Chapter3: Polynomial Functions
Section3.5: Mathematical Modeling And Variation
Problem 7ECP: The kinetic energy E of an object varies jointly with the object’s mass m and the square of the...
Related questions
Question
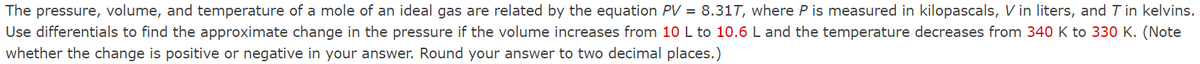
Transcribed Image Text:The pressure, volume, and temperature of a mole of an ideal gas are related by the equation PV = 8.31T, where P is measured in kilopascals, V in liters, and T in kelvins.
Use differentials to find the approximate change in the pressure if the volume increases from 10 L to 10.6 L and the temperature decreases from 340 K to 330 K. (Note
whether the change is positive or negative in your answer. Round your answer to two decimal places.)
Expert Solution
Step 1

Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you


Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage

College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning


Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage

College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning